205526-30-3
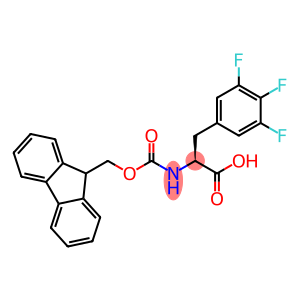
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-phenylalanine
CAS: 205526-30-3
Molecular Formula: C24H18F3NO4
205526-30-3 - Names and Identifiers
| Name | N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-phenylalanine |
| Synonyms | Fmoc-L-Phe(3,4,5-trifluoro)-OH Fmoc-L-3,4,5-Trifluorophenylalanine Fmoc-L-2-Amino-3-(3,4,5-trifluoro-phenyl)alanine (9H-Fluoren-9-yl)MethOxy]Carbonyl Phe(3,4,5-TriF)-OH N-ALPHA-(9-FLUORENYLMETHOXYCARBONYL)-3,4,5-TRIFLUORO-L-PHENYLALANINE N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-phenylalanine 2-[[9H-fluoren-9-ylmethoxy(oxo)methyl]amino]-3-(3,4,5-trifluorophenyl)propanoic acid (2S)-2-{[(9H-Fluoren-9-ylmethoxy)carbonyl]amino}-3-(3,4,5-trifluorophenyl)propanoic acid |
| CAS | 205526-30-3 |
| InChI | InChI=1/C24H18F3NO4/c25-19-9-13(10-20(26)22(19)27)11-21(23(29)30)28-24(31)32-12-18-16-7-3-1-5-14(16)15-6-2-4-8-17(15)18/h1-10,18,21H,11-12H2,(H,28,31)(H,29,30)/t21-/m1/s1 |
205526-30-3 - Physico-chemical Properties
| Molecular Formula | C24H18F3NO4 |
| Molar Mass | 441.4 |
| Density | 1.396g/cm3 |
| Boling Point | 616.5°C at 760 mmHg |
| Flash Point | 326.7°C |
| Vapor Presure | 4.66E-16mmHg at 25°C |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Refractive Index | 1.601 |
205526-30-3 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Hazard Class | IRRITANT |
205526-30-3 - Introduction
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-Phenylalanine is an organic compound with the following properties, uses, preparation and safety information:
Nature:
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4, 5-trifluororo-L-Phenylalanine is a white, crystalline solid that is flammable. It is stable at room temperature, but decomposes at high temperatures. Its chemical formula is C23H18F3NO4 and its relative molecular mass is 437.39.
Use:
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4, 5-trifluororo-l-phenylalanine is mainly used in the fields of biochemistry and organic synthesis, and is an important intermediate in peptide synthesis. It is often used in solid phase synthesis techniques to modify the N-terminus of a polypeptide or protein to increase its solubility and stability. In addition, it can also be used for the synthesis of biologically active peptide compounds.
Method:
The preparation of N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-Phenylalanine is generally carried out in the laboratory. A commonly used synthetic method is the reaction of Fmoc protected 3,4, 5-trifluorophenylalanine with the appropriate amine under appropriate conditions to yield the desired product. Care should be taken to use appropriate solvents, reagents and reaction conditions in the synthetic process.
Safety Information:
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4, 5-trifluororo-L-Phenylalanine is a chemical that requires proper safe handling procedures when used. It is irritating and may cause damage to the eyes, skin and respiratory system. Wear appropriate protective equipment, such as lab gloves, goggles and protective masks, when in use. At the same time, it should be stored in a dry, well-ventilated place, away from fire and oxidants. In case of accidental contact or inhalation, wash the affected area immediately and consult a doctor. Local laws and regulations should be followed when handling and disposing of waste.
Nature:
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4, 5-trifluororo-L-Phenylalanine is a white, crystalline solid that is flammable. It is stable at room temperature, but decomposes at high temperatures. Its chemical formula is C23H18F3NO4 and its relative molecular mass is 437.39.
Use:
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4, 5-trifluororo-l-phenylalanine is mainly used in the fields of biochemistry and organic synthesis, and is an important intermediate in peptide synthesis. It is often used in solid phase synthesis techniques to modify the N-terminus of a polypeptide or protein to increase its solubility and stability. In addition, it can also be used for the synthesis of biologically active peptide compounds.
Method:
The preparation of N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-Phenylalanine is generally carried out in the laboratory. A commonly used synthetic method is the reaction of Fmoc protected 3,4, 5-trifluorophenylalanine with the appropriate amine under appropriate conditions to yield the desired product. Care should be taken to use appropriate solvents, reagents and reaction conditions in the synthetic process.
Safety Information:
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4, 5-trifluororo-L-Phenylalanine is a chemical that requires proper safe handling procedures when used. It is irritating and may cause damage to the eyes, skin and respiratory system. Wear appropriate protective equipment, such as lab gloves, goggles and protective masks, when in use. At the same time, it should be stored in a dry, well-ventilated place, away from fire and oxidants. In case of accidental contact or inhalation, wash the affected area immediately and consult a doctor. Local laws and regulations should be followed when handling and disposing of waste.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: (S)-2-((((9H-Fluoren-9-yl)methoxy)carbonyl)amino)-3-(3,4,5-trifluorophenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 205526-30-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-phenylalanine Request for quotation
CAS: 205526-30-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 205526-30-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: FMOC-L-3,4,5-TRIFLUOROPHENYLALANINE Request for quotation
CAS: 205526-30-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 205526-30-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: FMOC-L-3,4,5-TRIFLUOROPHENYLALANINE Visit Supplier Webpage Request for quotation
CAS: 205526-30-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 205526-30-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-2-((((9H-Fluoren-9-yl)methoxy)carbonyl)amino)-3-(3,4,5-trifluorophenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 205526-30-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-3,4,5-trifluoro-L-phenylalanine Request for quotation
CAS: 205526-30-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 205526-30-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: FMOC-L-3,4,5-TRIFLUOROPHENYLALANINE Request for quotation
CAS: 205526-30-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 205526-30-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: FMOC-L-3,4,5-TRIFLUOROPHENYLALANINE Visit Supplier Webpage Request for quotation
CAS: 205526-30-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 205526-30-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



