204205-33-4
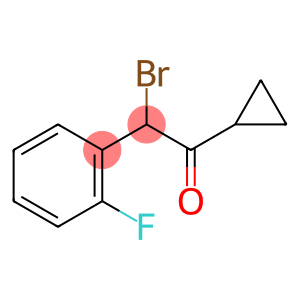
2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone
CAS: 204205-33-4
Molecular Formula: C11H10BrFO
204205-33-4 - Names and Identifiers
204205-33-4 - Physico-chemical Properties
| Molecular Formula | C11H10BrFO |
| Molar Mass | 257.1 |
| Density | 1.574±0.06 g/cm3(Predicted) |
| Boling Point | 293.0±25.0 °C(Predicted) |
| Flash Point | 131.009°C |
| Solubility | Chloroform (Sparingly), Ethyl Acetate (Slightly) |
| Vapor Presure | 0.002mmHg at 25°C |
| Appearance | Solid |
| Color | Yellow to Light Brown Oil to Low-Melting |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.592 |
204205-33-4 - Reference Information
| Introduction | 2-bromo-2-(2-fluorophenyl)-1-cyclopropylethyl ketone is a yellow or yellow-brown low melting point solid at room temperature and pressure, 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethyl ketone is a key synthetic intermediate of the drug prasugrel. |
| Use | 2-bromo-2-(2-fluorophenyl)-1-cyclopropylethyl ketone can be used as a pharmaceutical intermediate such as a synthetic intermediate of the drug molecule prasugrel. Pragre can prevent thrombosis in patients with coronary syndrome after percutaneous coronary intervention. Compared with the second-generation antiplatelet drug clopidogrel, it has the characteristics of faster, stronger, longer-lasting and safer effect. In the synthetic transformation, the bromine unit in the structure can be attacked by nucleophiles to obtain derivatized products; the ternary ring in the structure can undergo ring-opening functionalization reaction under the action of transition metals. |
| Synthesis method | Mix 150g cyclopropyl -2-fluorobenzyl ketone and 1.8 in a 10 liter 4-neck flask equipped with a condenser L methanol, then, add 147.9g of liquid bromine dropwise to the mixture within 3.5 hours, and further stir it at 25-30°C for 2.5 hours. Cool the mixture to 10°C, and add 3.8 l pre-cooled water drop by drop to the reactor, extract the reaction substance with 2.25 liters of ethyl acetate, separate the organic layer and the water layer, and the water layer is extracted with 750ml of ethyl acetate, Separate each layer in turn and mix the ethyl acetate layer and wash with 750ml 10% w/v sodium metabisulfite solution to separate each layer, the ethyl acetate layer was washed with 750ml 10% w/v sodium bicarbonate solution. The layers were separated, the ethyl acetate layer was washed with 750ml saline solution, the ethyl acetate layer was separated, dried with 100g sodium sulfate, and concentrated in vacuum at 50-55 ℃ to obtain the title compound. Yield: 94.65%,205 g, yellow oily liquid. the synthetic route of 2-bromo-2-(2-fluorophenyl)-1-cyclopropylethyl ketone |
Last Update:2024-04-09 02:00:07
Supplier List
Spot supply
Product Name: 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone Visit Supplier Webpage Request for quotationCAS: 204205-33-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone Request for quotation
CAS: 204205-33-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 204205-33-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone Visit Supplier Webpage Request for quotationCAS: 204205-33-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone Visit Supplier Webpage Request for quotationCAS: 204205-33-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone Request for quotation
CAS: 204205-33-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 204205-33-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: 2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone Visit Supplier Webpage Request for quotationCAS: 204205-33-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


