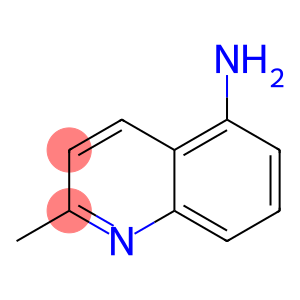
2-methylquinolin-5-amine
5-Aminoquinaldine
CAS: 54408-50-3
Molecular Formula: C10H10N2
2-methylquinolin-5-amine - Names and Identifiers
| Name | 5-Aminoquinaldine |
| Synonyms | AKOS 92235 AKOS BB-7774 ASISCHEM C75210 5-Aminoquinaldine AKOS BBS-00001935 CHEMBRDG-BB 4100117 2-METHYLQUINOLIN-5-AMINE 2-methylquinolin-5-amine 5-Amino-2-methylquinoline 5-QUINOLINAMINE, 2-METHYL- |
| CAS | 54408-50-3 |
| InChI | InChI=1/C10H10N2/c1-7-5-6-8-9(11)3-2-4-10(8)12-7/h2-6H,11H2,1H3 |
2-methylquinolin-5-amine - Physico-chemical Properties
| Molecular Formula | C10H10N2 |
| Molar Mass | 158.2 |
| Density | 1.169±0.06 g/cm3(Predicted) |
| Melting Point | 117-118°C |
| Boling Point | 316.6±27.0 °C(Predicted) |
| Flash Point | 171.1°C |
| Vapor Presure | 0.000405mmHg at 25°C |
| pKa | 6.39±0.50(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Refractive Index | 1.681 |
| Physical and Chemical Properties | melting point 117-118°C |
2-methylquinolin-5-amine - Reference Information
| Uses | 5-aminoquinaldine, also known as 5-amino-2-methylquinoline, is an important pharmaceutical intermediate. |
| preparation | CN201010559602.2 provides a simple process, mild conditions, simple post-treatment, high product purity, low cost and suitable for industrial production The preparation method of 5-amino-2-methylquinoline. The first step: preparation of 8-chloro -2-methylquinoline hydrochloride 1L reaction bottle add 50g o-chloroaniline, 130.5g n-butanol, 120g concentrated hydrochloric acid, 96.3g tetrachloroquinone, mechanical stirring, oil bath heating to 90-95 ℃; 33g crotonaldehyde and 33g n-butanol are evenly mixed, dripped into the reaction bottle, heated and refluxed for 1h after dripping. Cooling to 80 ℃, adding 53.4g of zinc chloride solid in batches, adding 240g of isopropyl ether, refluxing for 1h. Sampling analysis, reaction completed. Cool to 0 ℃, keep warm and stir for 1h, filter, and collect 83.1g of solid product. The purity of HPLC detection is ≥ 99%, and the yield is 99% (based on o-chloroaniline). step 2: preparation of 8-chloro -5-nitro -2-methylquinoline 558g concentrated sulfuric acid, g8-chloro -2-methylquinoline hydrochloride, 159.4 temperature control 35-45 ℃, dropwise addition of 99g fuming nitric acid (mass concentration 95%), after adding, stirring for 1h. Pour the reaction solution into ice water, adjust the pH to 9-10 with sodium hydroxide solution, filter, collect solids, and dry to obtain 164.1g of product. The purity of HPLC was ≥ 98%, and the yield was 99% (calculated as 8-chloro-2-methylquinoline hydrochloride). step 3: preparation of 5-amino -2-methylquinoline 87g 8-chloro -5-nitro -2-methylquinoline, 420g 95% ethanol, mechanical stirring, then 87.5g reduced iron powder, heating and reflux. Add 5.5g concentrated hydrochloric acid dropwise, add reflux reaction for 15h,TLC tracking. Without raw materials, filter while hot, wash the filter cake with a small amount of 95% ethanol, transfer the filtrate into a 1L single-mouth bottle, add 3.8g of 5% palladium carbon, and replace it with hydrogen. Then at 1 atmosphere, heat to 60-70 ℃ for 48 hours, sampling HPLC tracking. After the reaction is completed, 100g of water is added to the reaction mixture, heated and dissolved, filtered to remove palladium and carbon, the filtrate is concentrated to dry to obtain solid, dissolved in water, the pH of sodium hydroxide solution is adjusted to 9-10, filtered, solid is collected, and dried to obtain product. HPLC detection purity ≥ 98%, yield: 67%. |
| synthesis | in the prior art, it is reported in domestic and foreign literatures that aniline is mainly used as raw material, and then quinoline ring is synthesized with ethyl acetoacetate at a high temperature of 250 ℃, 5-nitro-2-methylquinoline is obtained by nitration, and finally the target product 5-amino -2-methylquinoline is obtained by catalytic hydrogenation. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 2-Methylquinolin-5-amine Visit Supplier Webpage Request for quotationCAS: 54408-50-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Aminoquinaldine Request for quotation
CAS: 54408-50-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 54408-50-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 5-Aminoquinaldine Request for quotation
CAS: 54408-50-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54408-50-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 5-Aminoquinaldine Visit Supplier Webpage Request for quotation
CAS: 54408-50-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 54408-50-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Methylquinolin-5-amine Visit Supplier Webpage Request for quotationCAS: 54408-50-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Aminoquinaldine Request for quotation
CAS: 54408-50-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 54408-50-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 5-Aminoquinaldine Request for quotation
CAS: 54408-50-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54408-50-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 5-Aminoquinaldine Visit Supplier Webpage Request for quotation
CAS: 54408-50-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 54408-50-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



