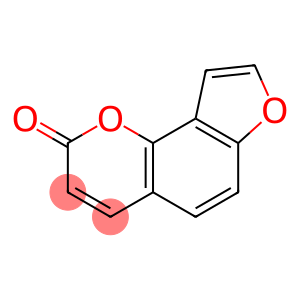
2-Oxo-(2H)-furo(2,3-h)-1-benzopyran
angelicin
CAS: 523-50-2
Molecular Formula: C11H6O3
2-Oxo-(2H)-furo(2,3-h)-1-benzopyran - Names and Identifiers
2-Oxo-(2H)-furo(2,3-h)-1-benzopyran - Physico-chemical Properties
| Molecular Formula | C11H6O3 |
| Molar Mass | 186.16 |
| Density | 1.2477 (rough estimate) |
| Melting Point | 132-134°C |
| Boling Point | 104°C/4mmHg(lit.) |
| Flash Point | 173.1°C |
| Solubility | Soluble in ethanol, chloroform, slightly soluble in water, ether and petroleum ether. |
| Vapor Presure | 1.91E-05mmHg at 25°C |
| Appearance | Colorless crystal |
| Color | White |
| Maximum wavelength(λmax) | ['302nm(EtOH)(lit.)'] |
| BRN | 153970 |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Sensitive | Sensitive to heat |
| Refractive Index | 1.6310 (estimate) |
| MDL | MFCD00064930 |
| Physical and Chemical Properties | White needle-like crystals, soluble in chloroform, derived from Psoralea corylifolia. |
2-Oxo-(2H)-furo(2,3-h)-1-benzopyran - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. R40 - Limited evidence of a carcinogenic effect |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S37/39 - Wear suitable gloves and eye/face protection S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| RTECS | LV0940000 |
| HS Code | 29322090 |
2-Oxo-(2H)-furo(2,3-h)-1-benzopyran - Reference
| Reference Show more | 1. Yang, Jie-Fang. Simultaneous determination of six main components in gukang capsules by LC-MS [J]. China pharmacy 2012 23(35):3344-3346. 2. Fan Binbing, Guo Yinhua, Huang Ting. Evaluation of processing technology of Psoralea corylifolia based on tissue distribution characteristics [J]. Journal of Liaoning University of Traditional Chinese Medicine, 2019, 21(02):58-60. 3. Xuanxian Jun, Mao Hongjiao, Song Bingjie, etc. Inhibitory effect of isopsoralen on osteolysis induced by tricalcium phosphate granules [J]. Chinese Journal of Nutrition, 2016, 38(003):273-277. 4. Zhang Na, Liu Chuanxin, Zhang chenning, etc. Mechanism of Hepatotoxicity induced by Psoralea corylifolia L. Combined with UPLC-QTOF-MS and network toxicology techniques [J]. International Journal of Pharmaceutical Research, Vol. 47, No. 5, 2020, pp. 370-376, ISTIC PKU CSCD, 2020. 5. Ding Yuting, Zheng Zhuhong, Zhao renyun, etc. UPLC-Q-TOF-MS ~ E technology combined with UNIFI screening platform for rapid analysis of chemical constituents of Psoralea corylifolia [J]. Journal of Mass Spectrometry 2018. 6. Cao Shuli Zhao Chunjie Cai Li Xia Jing Minqi Li Yibai. Simultaneous determination of psoralen, isopsoralen, atractylenolide Ⅲ and isoimperatorin in Shenling Baizhu powder by UPLC [J]. Journal of Shenyang Pharmaceutical University 2018 35(07):568-573. 7. Zheng Nan, Guo Yihua, Liu Min, et al. Study on fingerprint of different processed products of Psoralea corylifolia L. [J]. Liaoning Journal of Traditional Chinese Medicine, 2015(04):157-159. 8. Li, Yan, Zhou, Lu, Wang, Lina, et al. Determination of content of different types of components in Psoralea corylifolia L. By one-Test multi-evaluation method [J]. Chinese Journal of Experimental prescriptions, 2013, 19(5):152-154. 9. Huang Ting, Guo Yuhua, Ding Liyan, etc. Effect of Psoralea corylifolia L. Juice on anti-OP chemical composition [J]. Journal of Liaoning University of Traditional Chinese Medicine, 2013, 22 (05):62-64. 10. Xuelian Wang, Minmin Li, Junhua Hu, et al. Inhibitory effects of 18 coumarins on Colletotrichum gloeosporioides in Citrus [J]. Southern China fruit trees, 2013, 42(004):73-74. 11. Li Jiayan, Liu Xinxin, Ning Qing, Li Yandong, Shi Ziqi, Wei Yingjie. Efficient Identification of toxicity related components of Psoralea corylifolia based on zebrafish model [J]. Chinese herbal medicine, 2021,52(01):129-136. 12. Chen, Zhi-Jing, et al. "Dietary total prenylflavonoids from the fruits of Psoralea corylifolia L. prevents age-related cognitive deficits and down-regulates Alzheimer's markers in SAMP8 mice." Molecules 23.1 (2018): 196.https://doi.org/10.3390/molecules230 13. Chen, Xiumin, Yanfang Yang, and Yingtao Zhang. "Isobavachalcone and bavachinin from Psoraleae Fructus modulate Aβ42 aggregation process through different mechanisms in vitro." Febs Letters 587.18 (2013): 2930-2935.https://doi.org/10.1016/j.febslet.2013.07. 14. [IF=4.411] Li Jia et al."Systematic Profiling of the Multicomponents and Authentication of Erzhi Pill by UHPLC/Q-Orbitrap-MS Oriented Rapid Polarity-Switching Data-Dependent Acquisition and Selective Monitoring of the Chemical Markers Deduced from Fingerprint Analys 15. [IF=2.675] Xiumin Chen et al."Isobavachalcone and bavachinin from Psoraleae Fructus modulate Aβ42 aggregation process through different mechanisms in vitro."Febs Lett. 2013 Sep;587(18):2930-2935 16. [IF=1.908] Dan Lu et al."Mechanism-based inactivation of cytochrome P450 2B6 by isopsoralen."Xenobiotica. 2016;46(4):335-341 17. [IF=5.455] Zhang Xian et al."Neurobehavioral alternations of the female offspring born to polycystic ovary syndrome model rats administered by Chinese herbal medicine."Chin Med-Uk. 2021 Dec;16(1):1-19 18. [IF=3] Qu Lala et al."Phenotypic assessment and ligand screening of ETA/ETB receptors with label-free dynamic mass redistribution assay."N-S Arch Pharmacol. 2020 Jun;393(6):937-950 |
2-Oxo-(2H)-furo(2,3-h)-1-benzopyran - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| (IARC) carcinogen classification | 3 (Vol. 40, Sup 7) 1987 |
| introduction | isopsoralen is a furanocoumarin, usually isolated from the seeds of p corylifolia. Like other furanocoumarins, isopsoralen has antibacterial activity against many Gram (+) and Gram (-) bacteria. It was also shown to prevent tacrine-induced cytotoxicity in human liver-derived HepG2 cells (EC50=47 μg/ml) and vascular relaxation in the aorta of rats precontracted by phenylephrine. |
| toxicity | the LD50 of isopsoralen is 322 mg/kg, which is acute toxicity to rats orally. Possible side effects include changing circadian rhythm and rerection, ataxia, and analgesia. |
| biological activity | isopsoralen (Angelicin) is a natural tricyclic aromatic hydrocarbon compound, which is structurally related to psoralen and has anti-cancer, Anti-inflammatory, antiviral and other activities. Cytotoxic, IC50:49.56 μM; Inhibitory MHV-68,IC50: 5.39 μg/ml (28.95 μM). |
| use | isopsoralen has antibacterial and anti-inflammatory effects. used for content determination/identification/pharmacological experiment, etc. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 21:54:55
Supplier List
Product Name: angelicin Request for quotation
CAS: 523-50-2
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
CAS: 523-50-2
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
Spot supply
Product Name: angelicin Request for quotationCAS: 523-50-2
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Spot supply
Product Name: Angelicin Visit Supplier Webpage Request for quotationCAS: 523-50-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: Angelicin Visit Supplier Webpage Request for quotationCAS: 523-50-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Isopsoralen Visit Supplier Webpage Request for quotationCAS: 523-50-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: angelicin Request for quotation
CAS: 523-50-2
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
CAS: 523-50-2
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
Spot supply
Product Name: angelicin Request for quotationCAS: 523-50-2
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Spot supply
Product Name: Angelicin Visit Supplier Webpage Request for quotationCAS: 523-50-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: Angelicin Visit Supplier Webpage Request for quotationCAS: 523-50-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Isopsoralen Visit Supplier Webpage Request for quotationCAS: 523-50-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




