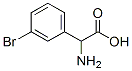
2-AMINO-2-(3-BROMOPHENYL)ACETIC
Amino(3-bromophenyl)acetic acid
CAS: 79422-73-4
Molecular Formula: C8H8BrNO2
2-AMINO-2-(3-BROMOPHENYL)ACETIC - Names and Identifiers
2-AMINO-2-(3-BROMOPHENYL)ACETIC - Physico-chemical Properties
| Molecular Formula | C8H8BrNO2 |
| Molar Mass | 230.06 |
| Density | 1.673±0.06 g/cm3(Predicted) |
| Melting Point | 212-215° |
| Boling Point | 351.6±32.0 °C(Predicted) |
| Flash Point | 166.4°C |
| Vapor Presure | 1.51E-05mmHg at 25°C |
| pKa | 1.76±0.10(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.624 |
2-AMINO-2-(3-BROMOPHENYL)ACETIC - Risk and Safety
| Hazard Class | IRRITANT |
2-AMINO-2-(3-BROMOPHENYL)ACETIC - Introduction
Amino(3-bromophenyl)acetic acid, also known as bromophenylglycine or amino (3-bromophenyl)acetic acid, is an organic compound. The following is a description of the nature, use, formulation and safety information of Amino(3-bromophenyl)acetic acid:
properties: Amino(3-bromophenyl)acetic acid has a chemical formula of C8H8BrNO3 and a molecular weight of 240.06g/mol. It is a white crystalline solid, flammable, easily soluble in water and some organic solvents. Under acidic conditions, Amino(3-bromophenyl) acitic acid may produce toxic hydrogen bromide gas.
Uses: Amino(3-bromophenyl)acetic acid has some uses in chemical and biological research. It is useful as an intermediate in organic synthesis, for example in the synthesis of pharmaceuticals or other organic compounds. In addition, it can be used for the synthesis of biologically active molecules and materials, for example for the design of novel drugs or dyes.
Preparation method: Amino(3-bromophenyl)acetic acid can be prepared by reacting phenylglycine with bromide. Specific synthetic routes need to be carried out under appropriate laboratory conditions and require the use of appropriate experimental techniques and tools.
Safety information: The following safety precautions should be noted for the use and operation of Amino(3-bromophenyl)acetic acid:
1. Use should follow appropriate laboratory safety procedures, such as wearing protective glasses, laboratory gloves and laboratory coats.
2. Since Amino(3-bromophenyl)acetic acid can cause the release of hydrogen bromide gas, it should be operated under sufficient ventilation conditions.
3. Avoid contact with skin, eyes and respiratory tract. In case of contact, flush the affected area immediately with plenty of water and seek medical attention immediately.
4. storage should be sealed and stored in a dry, cool place.
5. Local environmental regulations should be followed when handling and disposing of waste.
properties: Amino(3-bromophenyl)acetic acid has a chemical formula of C8H8BrNO3 and a molecular weight of 240.06g/mol. It is a white crystalline solid, flammable, easily soluble in water and some organic solvents. Under acidic conditions, Amino(3-bromophenyl) acitic acid may produce toxic hydrogen bromide gas.
Uses: Amino(3-bromophenyl)acetic acid has some uses in chemical and biological research. It is useful as an intermediate in organic synthesis, for example in the synthesis of pharmaceuticals or other organic compounds. In addition, it can be used for the synthesis of biologically active molecules and materials, for example for the design of novel drugs or dyes.
Preparation method: Amino(3-bromophenyl)acetic acid can be prepared by reacting phenylglycine with bromide. Specific synthetic routes need to be carried out under appropriate laboratory conditions and require the use of appropriate experimental techniques and tools.
Safety information: The following safety precautions should be noted for the use and operation of Amino(3-bromophenyl)acetic acid:
1. Use should follow appropriate laboratory safety procedures, such as wearing protective glasses, laboratory gloves and laboratory coats.
2. Since Amino(3-bromophenyl)acetic acid can cause the release of hydrogen bromide gas, it should be operated under sufficient ventilation conditions.
3. Avoid contact with skin, eyes and respiratory tract. In case of contact, flush the affected area immediately with plenty of water and seek medical attention immediately.
4. storage should be sealed and stored in a dry, cool place.
5. Local environmental regulations should be followed when handling and disposing of waste.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 2-Amino-2-(3-bromophenyl)acetic acid Visit Supplier Webpage Request for quotationCAS: 79422-73-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINO-2-(3-BROMOPHENYL)ACETIC ACID Request for quotation
CAS: 79422-73-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 79422-73-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-Amino-2-(3-bromophenyl)acetic Acid Visit Supplier Webpage Request for quotation
CAS: 79422-73-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 79422-73-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Amino-2-(3-bromophenyl)acetic acid Visit Supplier Webpage Request for quotationCAS: 79422-73-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINO-2-(3-BROMOPHENYL)ACETIC ACID Request for quotation
CAS: 79422-73-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 79422-73-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-Amino-2-(3-bromophenyl)acetic Acid Visit Supplier Webpage Request for quotation
CAS: 79422-73-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 79422-73-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


