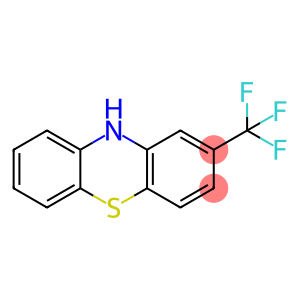
2-(trifluoromethyl)-10H-phenothiazine
2-(Trifluoromethyl)phenothiazine
CAS: 92-30-8
Molecular Formula: C13H8F3NS
2-(trifluoromethyl)-10H-phenothiazine - Names and Identifiers
| Name | 2-(Trifluoromethyl)phenothiazine |
| Synonyms | TIMTEC-BB SBB003058 Trifluoromethylphenothiazine 2-trifluoromethylphenothiazine 2-(trifluoromethyl)-phenothiazin 2-(Trifluoromethyl)phenothiazine 2-Trilfluoromethyl Phenothiazine 2-(TRIFLOUOMETHYL) PHENOTHIAZINE 2-(Trifluoromethyl)dibenzothiazine Phenothiazine, 2-(trifluoromethyl)- 2-(Trifluoromethyl)thiodiphenylamine 2-(trifluoromethyl)-10H-phenothiazine 10H-Phenothiazine, 2-(trifluoromethyl)- (2R,5R)-2,5-bis(2,4,5-trimethoxyphenyl)tetrahydrofuran |
| CAS | 92-30-8 |
| EINECS | 202-145-7 |
| InChI | InChI=1/C22H28O7/c1-23-17-11-21(27-5)19(25-3)9-13(17)15-7-8-16(29-15)14-10-20(26-4)22(28-6)12-18(14)24-2/h9-12,15-16H,7-8H2,1-6H3/t15-,16-/m1/s1 |
| InChIKey | RKGYJVASTMCSHZ-UHFFFAOYSA-N |
2-(trifluoromethyl)-10H-phenothiazine - Physico-chemical Properties
| Molecular Formula | C13H8F3NS |
| Molar Mass | 267.27 |
| Density | 1.3491 (estimate) |
| Melting Point | 188-190°C(lit.) |
| Boling Point | 361.1±42.0 °C(Predicted) |
| Flash Point | 210°C |
| Solubility | DMSO (Slightly), Methanol (Slightly) |
| Vapor Presure | 1.49E-10mmHg at 25°C |
| Appearance | Powder |
| Color | Pale Yellow to Light Green |
| BRN | 226580 |
| pKa | -2.60±0.20(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.531 |
| MDL | MFCD00005018 |
| Physical and Chemical Properties | Appearance: light yellow green powder Melting Point: 190-194°C |
2-(trifluoromethyl)-10H-phenothiazine - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R22 - Harmful if swallowed |
| Safety Description | S36 - Wear suitable protective clothing. S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | SP5620000 |
| TSCA | T |
| HS Code | 29349990 |
| Hazard Note | Irritant |
2-(trifluoromethyl)-10H-phenothiazine - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Application | 2-trifluoromethylphenothiazine is an organic intermediate, 3-trifluoromethyldiphenylamine can be first prepared from flufenamic acid, and 2-trifluoromethylphenothiazine can be obtained by how to close the ring. |
| preparation | preparation of 2-trifluoromethylphenothiazine: add 100g(0.356mol) of flufenamic acid to the reaction flask, after it is completely melted, 10g(0.178mol) of iron powder is added to the reaction flask, and the reaction is stirred at a temperature of 180-190 ° C. For about 2 hours, when the reaction solution was cooled to 100 ° C., the reaction solution was poured into a beaker while hot, and the iron powder was left in the bottom of the reaction flask (which could be rinsed with water). The reaction solution was added to a clean reaction flask and distilled under reduced pressure to collect a 134-135 ° C. (3mmHg) fraction to obtain about 67.5g of 3-trifluoromethyldiphenylamine as a pale yellow liquid in a yield of about 80%. 60g(0.253mol) of 3-trifluoromethyldiphenylamine and 8g(0.253mol) of Shenghua sulfur are added into the reaction flask, and the mixture is stirred and heated to about 130 ℃, after sulfur is completely melted, 3g of iodine is added to the reaction flask, The temperature was continued to 185-190 °c and the reaction was carried out at this temperature for about 1 hour. Hydrogen sulfide gas is released during the reaction, and the absorption of tail gas should be paid attention. After completion of the reaction, the reaction solution is cooled to about 100 °c, 200g of toluene is added to the reaction flask, the temperature is raised to about 100 °c, g of water is added to the reaction flask, stirred for 5 minutes, and then layered while hot, the water layer was discarded, and the toluene layer was returned to the reaction bottle, cooled to 15-18 ℃ under stirring, filtered, and the filtrate was retained (3-trifluoromethyldiphenylamine was to be used for recovery), the filter cake was dried under vacuum at 60 °c for 10 hours to obtain 29g of the intermediate 2-trifluoromethylphenothiazine in about 85% yield. |
| Use | an intermediate of trifluoperazine, fluphenazine. |
| production method | 2-carboxy-3-(trifluoromethyl) diphenylamine (I. E., a synthetic antibacterial agent, flufenamic acid) by high temperature catalytic decarboxylation (200 deg C, iron powder), the formation of 3-(trifluoromethyl) diphenylamine. The latter is heated together with sulfur in the presence of iodine and cyclized to produce the product. |
Last Update:2024-04-09 15:16:53
Supplier List
Spot supply
Product Name: 2-(Trifluoromethyl)-10H-phenothiazine Visit Supplier Webpage Request for quotationCAS: 92-30-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-(Trifluoromethyl)phenothiazine Request for quotation
CAS: 92-30-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 92-30-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-(Trifluoromethyl)phenothiazine Request for quotation
CAS: 92-30-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 92-30-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-(Trifluoromethyl)-Phenothiazine Visit Supplier Webpage Request for quotationCAS: 92-30-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-(Trifluoromethyl)-10H-phenothiazine Visit Supplier Webpage Request for quotationCAS: 92-30-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-(Trifluoromethyl)phenothiazine Request for quotation
CAS: 92-30-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 92-30-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-(Trifluoromethyl)phenothiazine Request for quotation
CAS: 92-30-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 92-30-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-(Trifluoromethyl)-Phenothiazine Visit Supplier Webpage Request for quotationCAS: 92-30-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



