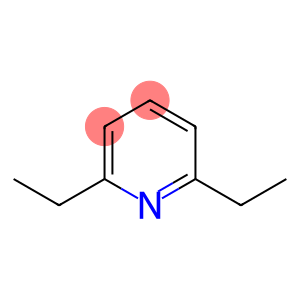
2,6-Diethyl-pyridine
2,6-diethylpyridine
CAS: 935-28-4
Molecular Formula: C9H13N
2,6-Diethyl-pyridine - Names and Identifiers
| Name | 2,6-diethylpyridine |
| Synonyms | 2,6-diethylpyridine 2,6-Diethyl-pyridine Pyridine, 2,6-diethyl- |
| CAS | 935-28-4 |
| InChI | InChI=1/C9H13N/c1-3-8-6-5-7-9(4-2)10-8/h5-7H,3-4H2,1-2H3 |
2,6-Diethyl-pyridine - Physico-chemical Properties
| Molecular Formula | C9H13N |
| Molar Mass | 135.21 |
| Density | 0.9354 (estimate) |
| Melting Point | 31.33°C (estimate) |
| Boling Point | 203.93°C (estimate) |
| Flash Point | 54.5°C |
| Vapor Presure | 1.46mmHg at 25°C |
| pKa | 6.56±0.10(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.4976 (estimate) |
2,6-Diethyl-pyridine - Introduction
2,6-diethylpyriridine is an organic compound with the chemical formula C10H15N. It is a colorless liquid with a special smell. The following is a description of the nature, use, preparation and safety information of 2,6-diethylpyriridine:
Nature:
-Appearance: colorless liquid
-Molecular weight: 161.24g/mol
-Density: 0.87g/cm³
-Boiling point: 183-186 ℃
-Melting point:-93 ℃
-Solubility: Slightly soluble in water, soluble in most organic solvents
Use:
- 2,6-diethylpyrridine is commonly used as an intermediate in organic synthesis.
-It can be used as an ingredient in pharmaceutical synthesis, rubber processing aids, and certain coatings and adhesives.
-Due to its alkalinity, it can also be used as a catalyst or coordination reagent.
Method:
-2,6-diethylpyrridine is usually synthesized by adding ethyl halide or ethyl compound to pyridine.
-Ethyl chloride, ethyl bromide, ethyl sulfonate, or the like can be used as an ethylating agent.
-This reaction is generally carried out under basic conditions, for example in a basic solvent such as sodium glycol solution.
Safety Information:
- 2,6-diethylpyrridine is irritating and may cause irritation to the eyes, skin and respiratory tract.
-Avoid direct contact with skin and eyes when using, and ensure adequate ventilation.
-Wear appropriate protective equipment such as gloves, goggles and masks during operation.
-In case of contact with skin or eyes, rinse immediately with plenty of water and seek medical help if necessary.
Please note that the above information is for reference only. Please follow the relevant safety operation guidelines and laboratory regulations when using it in practice.
Nature:
-Appearance: colorless liquid
-Molecular weight: 161.24g/mol
-Density: 0.87g/cm³
-Boiling point: 183-186 ℃
-Melting point:-93 ℃
-Solubility: Slightly soluble in water, soluble in most organic solvents
Use:
- 2,6-diethylpyrridine is commonly used as an intermediate in organic synthesis.
-It can be used as an ingredient in pharmaceutical synthesis, rubber processing aids, and certain coatings and adhesives.
-Due to its alkalinity, it can also be used as a catalyst or coordination reagent.
Method:
-2,6-diethylpyrridine is usually synthesized by adding ethyl halide or ethyl compound to pyridine.
-Ethyl chloride, ethyl bromide, ethyl sulfonate, or the like can be used as an ethylating agent.
-This reaction is generally carried out under basic conditions, for example in a basic solvent such as sodium glycol solution.
Safety Information:
- 2,6-diethylpyrridine is irritating and may cause irritation to the eyes, skin and respiratory tract.
-Avoid direct contact with skin and eyes when using, and ensure adequate ventilation.
-Wear appropriate protective equipment such as gloves, goggles and masks during operation.
-In case of contact with skin or eyes, rinse immediately with plenty of water and seek medical help if necessary.
Please note that the above information is for reference only. Please follow the relevant safety operation guidelines and laboratory regulations when using it in practice.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 2,6-Diethylpyridine Visit Supplier Webpage Request for quotationCAS: 935-28-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,6-diethylpyridine Request for quotation
CAS: 935-28-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 935-28-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 2,6-diethylpyridine Visit Supplier Webpage Request for quotation
CAS: 935-28-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 935-28-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2,6-Diethylpyridine Visit Supplier Webpage Request for quotationCAS: 935-28-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,6-diethylpyridine Request for quotation
CAS: 935-28-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 935-28-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 2,6-diethylpyridine Visit Supplier Webpage Request for quotation
CAS: 935-28-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 935-28-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


