2,4-Difluorotolune
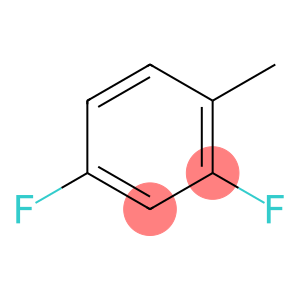
2,4-Difluorotoluene
CAS: 452-76-6
Molecular Formula: C7H6F2
2,4-Difluorotolune - Names and Identifiers
| Name | 2,4-Difluorotoluene |
| Synonyms | Difluorotoluene 2,4-Difluorotolune 2,4-DIFLUOROTOLUNE 2,4-Difluorotoluene 2,4-DIFLUOROTOLUENE 2,4-DIFLUOROTOLUINE 2.4-Difluoro methyl benzene 1-Methyl-2,4-difluorobenzene 2,4-DIFLUORO-1-METHYL-BENZENE Benzene, 2,4-difluoro-1-methyl- |
| CAS | 452-76-6 |
| EINECS | 207-211-9 |
| InChI | InChI=1/C7H8FN/c1-5-2-3-6(9)4-7(5)8/h2-4H,9H2,1H3 |
2,4-Difluorotolune - Physico-chemical Properties
| Molecular Formula | C7H6F2 |
| Molar Mass | 128.12 |
| Density | 1.12 g/mL at 25 °C (lit.) |
| Melting Point | -35 °C |
| Boling Point | 113-117 °C (lit.) |
| Flash Point | 59°F |
| Vapor Presure | 0.272mmHg at 25°C |
| Appearance | Liquid |
| Specific Gravity | 1.120 |
| Color | Clear colorless |
| BRN | 1931681 |
| Storage Condition | Sealed in dry,2-8°C |
| Refractive Index | n20/D 1.449(lit.) |
| Physical and Chemical Properties | Boiling Point: 114 - 116 density: 1.15 flash point: 13 |
2,4-Difluorotolune - Risk and Safety
| Risk Codes | 11 - Highly Flammable |
| Safety Description | S16 - Keep away from sources of ignition. S29 - Do not empty into drains. S33 - Take precautionary measures against static discharges. |
| UN IDs | UN 1993 3/PG 2 |
| WGK Germany | 3 |
| HS Code | 29039990 |
| Hazard Note | Flammable |
| Hazard Class | 3 |
| Packing Group | II |
2,4-Difluorotolune - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| Application | 2,4-difluorotoluene is an organic intermediate that can be prepared from 2,4-diaminotoluene through diazotization reaction. There are reports that it can be used to prepare 2, 4-difluorobenzaldehyde. |
| preparation | in a 250ml three-neck bottle, 2, 4-diaminotoluene (6.1g,0.05mol) is added, concentrated hydrochloric acid (36ml) is added, 40% fluoroboric acid (32ml) is added under ice salt bath cooling and stirring, and sodium nitrite (7.6g,0.11mol) solution dissolved in water (12ml). After adding, stir for 1h, filter, and wash the solid with ice water (20ml × 2), anhydrous ethanol and anhydrous ether (15ml × 2 each) respectively. Put into a calcium chloride dryer and dry for 2 hours. Add the diazonium salt (10g) obtained above into a 150ml round bottom flask, carefully heat with an alcohol lamp, and carry out thermal decomposition until a large amount of smoke is generated in the flask, and always maintain above this temperature until no smoke is generated, and the overflow is cooled by a straight condenser tube and a multi-stage cold trap, and the receiving tube is inserted into a conical flask containing ether. After the reaction, it was extracted with ether, combined with ether solution, added anhydrous calcium chloride for drying, and fractionated to obtain 2,4-difluorotoluene (5.3g, yield 82.8%),bp113~117 ℃ (literature: bp112 ℃/99.06kPa, yield 57%). |
| uses | intermediates in medicine, pesticides and liquid crystal materials. |
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: 2,4-Difluorotoluene Visit Supplier Webpage Request for quotationCAS: 452-76-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4-Difluorobiphenyl Request for quotation
CAS: 452-76-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 452-76-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2,4-Difluorotoluene Request for quotation
CAS: 452-76-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 452-76-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2,4-Difluorotoluene Visit Supplier Webpage Request for quotationCAS: 452-76-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4-Difluorobiphenyl Request for quotation
CAS: 452-76-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 452-76-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2,4-Difluorotoluene Request for quotation
CAS: 452-76-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 452-76-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
View History


