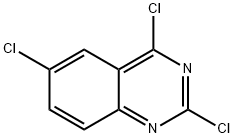
2,4,6-Trichloroquinazoline
2,4,6-Trichloroquinazoline
CAS: 20028-68-6
Molecular Formula: C8H3Cl3N2
2,4,6-Trichloroquinazoline - Names and Identifiers
| Name | 2,4,6-Trichloroquinazoline |
| Synonyms | 2,4,6-trichlroquinazoline 2,4,6-TRICHLOROQUINAZOLINE 2,4,6-Trichloroquinazoline 2,4,6-TRICHLOROQUINOXALINE Quinazoline, 2,4,6-trichloro- quinazoline, 2,4,6-trichloro- |
| CAS | 20028-68-6 |
| InChI | InChI=1/C8H3Cl3N2/c9-4-1-2-6-5(3-4)7(10)13-8(11)12-6/h1-3H |
2,4,6-Trichloroquinazoline - Physico-chemical Properties
| Molecular Formula | C8H3Cl3N2 |
| Molar Mass | 233.48 |
| Density | 1.600±0.06 g/cm3(Predicted) |
| Melting Point | 131 °C |
| Boling Point | 230-240 °C(Press: 15 Torr) |
| Flash Point | 162.566°C |
| Vapor Presure | 0.002mmHg at 25°C |
| pKa | -1.15±0.30(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.678 |
2,4,6-Trichloroquinazoline - Risk and Safety
| Risk Codes | R25 - Toxic if swallowed R37/38 - Irritating to respiratory system and skin. R41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
2,4,6-Trichloroquinazoline - Reference Information
| Introduction | quinazoline is an important class of benzo-heterocyclic compounds containing two nitrogen atoms, its derivatives have a variety of biological activities, such as anti-malaria, anti-fungal, anti-inflammatory, anti-hypertension, hypoglycemic, anti-tumor and anti-HIV activity. Therefore, the synthesis of quinazoline derivatives has received extensive attention. 2, 4, 6-trichloroquinazoline is an important quinazoline derivatives, can be prepared by a variety of synthetic routes, for example, 2, 4, 6-trichloroquinazoline is prepared by using 2-amino-5-chlorobenzoic acid as a starting material through cyclization reaction and chlorination reaction. |
| Use | 2,4, 6-trichloroquinazoline is a heterocyclic organic compound and can be used as a pharmaceutical intermediate. |
| preparation | 2, 4-dichloroquinazoline derivatives are synthesized 2, an important intermediate of 4-disubstituted quinazoline, via nucleophilic substitution reactions or transition metal-catalyzed coupling reactions, A variety of functional groups can be introduced at the 2-and 4-positions of the quinazoline ring. The synthesis of 2, 4-dichloroquinazoline derivatives is generally obtained by chloro reaction of quinazoline 2, 4-dione. There are many synthetic methods of quinazoline 2, 4-dione, such as: the condensation of anthranilic acid with excess urea at 150 ℃ ~ 200 ℃, condensation of anthranilic acid derivatives with cyanates under acidic conditions, reaction of anthranilic acid amide with phosgene, reaction of anthranilic acid acetonitrile with carbon dioxide in 1, 8-diazacyclo-bicyclo [5, 4, catalytic condensation of 0] -7-undecene, etc. However, the above method has the disadvantages of large amount of urea, harsh reaction conditions, long reaction time or low reaction yield. Recently, Lee et al used O-aminophenylacetonitrile and diphosgene reaction, using acetonitrile as solvent, in a sealed tube pressure reaction for 12 hours, one-step reaction synthesis of 2, 4-dichloroquinazoline derivative [1]. However, the application of this method is greatly limited due to the great toxicity of diphosgene. In this paper, phosphorus oxychloride (POCl3) as solvent, in the presence of phosphorus pentachloride ( PCl5), 2-amino-5-chlorobenzoic acid and urea condensation, one-pot reaction directly synthesized 2, 4, 6-trichloroquinazoline. The synthesis reaction scheme is shown in the following figure. Fig.1 reaction formula for synthesis of 2, 4, 6-trichloroquinazoline |
Last Update:2024-04-09 21:50:46
Supplier List
Spot supply
Product Name: 2,4,6-Trichloroquinazoline Visit Supplier Webpage Request for quotationCAS: 20028-68-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,6-TRICHLOROQUINAZOLINE Request for quotation
CAS: 20028-68-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 20028-68-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: 2,4,6-Trichloroquinazoline Visit Supplier Webpage Request for quotationCAS: 20028-68-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,6-TRICHLOROQUINAZOLINE Request for quotation
CAS: 20028-68-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 20028-68-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History

