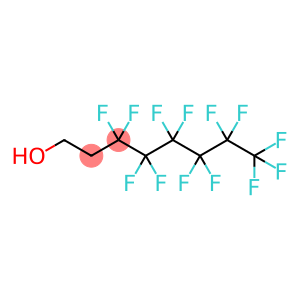
1H,1H,2H,2H-Perfluorooctanol
3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluorooctan-1-ol
CAS: 647-42-7
Molecular Formula: C8H5F13O
1H,1H,2H,2H-Perfluorooctanol - Names and Identifiers
| Name | 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluorooctan-1-ol |
| Synonyms | Capstone 62-AL 2-(Perfluorohexyl)ethanol 1H,1H,2H,2H-Perfluorooctanol 1H,1H,2H,2H-PERFLUOROOCTANOL 1H,1H,2H,2H-PERFLUOROOCTAN-1-OL 1H,1H,2H,2H-PERFLUORO-1-OCTANOL 1,1,2,2-Tetrahydroperfluoro-1-octanol 1H,1H,2H,2H-Tridecafluoro-1-n-octanol 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluoro-1-octano 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluorooctan-1-ol 3,3,4,4,5,5,6,6,7,7,8,8,8-Tridecafluoro-1-octanol 3,3,4,4,5,5,6,6,7,7,8,8,8-Tridecafluoro-octan-1-ol 1-Octanol, 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluoro- |
| CAS | 647-42-7 |
| EINECS | 211-477-1 |
| InChI | InChI=1/C8H5F13O/c9-3(10,1-2-22)4(11,12)5(13,14)6(15,16)7(17,18)8(19,20)21/h22H,1-2H2 |
| InChIKey | GRJRKPMIRMSBNK-UHFFFAOYSA-N |
1H,1H,2H,2H-Perfluorooctanol - Physico-chemical Properties
| Molecular Formula | C8H5F13O |
| Molar Mass | 364.1 |
| Density | 1.651g/mLat 25°C(lit.) |
| Boling Point | 88-95°C28mm Hg(lit.) |
| Flash Point | 197°F |
| Water Solubility | Miscible with chloroform and methanol. Immiscible with water. |
| Solubility | Chloroform (Sparingly), Methanol (Sparingly) |
| Vapor Presure | 0.382mmHg at 25°C |
| Appearance | liquid |
| Specific Gravity | 1.680 |
| Color | colorless |
| BRN | 1992757 |
| pKa | 14.26±0.10(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.313(lit.) |
1H,1H,2H,2H-Perfluorooctanol - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection S24/25 - Avoid contact with skin and eyes. |
| UN IDs | NA 1993 / PGIII |
| WGK Germany | 1 |
| RTECS | RH1460000 |
| TSCA | T |
| HS Code | 29055900 |
| Hazard Class | IRRITANT |
1H,1H,2H,2H-Perfluorooctanol - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Overview | perfluoroalkyl ethyl (meth) acrylate is an important fluorinated chemical, its polymer has excellent oil resistance and water resistance, is an important intermediate for the production of surface protective agents, and is widely used in textile, coating and fluorinated surfactant fields. However, perfluorooctanoic acid (PFOA) is difficult to be excreted in vitro because of its biological accumulation and persistence, and (meth) acrylate or its polymer containing perfluorooctyl (C8) is decomposed, oxidation, etc., and finally PFOA was generated. |
| Introduction | 3,3,4,4,5,5,6,6,7,7,8,8, 8-trifluoro-1-octanol, also known as perfluorohexyl ethanol, is a perfluoroalkyl ethanol. Perfluoroalkyl ethanol is an important intermediate for the synthesis of perfluoroalkyl surfactants and functional materials, because of its unique chemical stability, corrosion resistance, flame retardant, hydrophobic and oleophobic, antifouling and so on are widely used in textile, military, electronics, aerospace, anti-corrosion, chemical and other fields, and its preparation method is a research hotspot in recent years. |
| Use | perfluorohexyl ethyl alcohol is an intermediate for the preparation of perfluorohexyl ethyl acrylate. perfluorohexylethyl alcohol is a material used to improve nanotube composites. It can also be used in the synthesis of recycled fluorhydrazino thiosulfate compounds with NCS to catalyze the acetalization of aldehydes. |
| prepare | A 5L glass reaction bottle with Condenser tube, magnetic stirrer and thermometer, 474G of perfluorohexyl ethyl iodide (C6F13CH2CH2I) was added, 2370g of mixed solvent (nmp1896 G, DMSO 474g), and 108g of water. The reaction was stirred and heated to 160 ℃ for 18H. Gas phase detection also contained perfluorohexyl ethyl iodide 1.8%, 4,5,5,6,6,7,7,8,8, 8-tridecafluoro-1-octanol, conversion rate 99.3%, yield 96.0%, gas chromatography purity 99.4%. |
Last Update:2024-04-09 21:01:54
Supplier List
Featured ProductsMultiple SpecificationsSpot supply
Product Name: 1H,1H,2H,2H-PERFLUORO-1-OCTANOL Request for quotationCAS: 647-42-7
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Spot supply
Product Name: 1H,1H,2H,2H-Perfluorooctanol Visit Supplier Webpage Request for quotationCAS: 647-42-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1H,1H,2H,2H-PERFLUORO-1-OCTANOL Request for quotation
CAS: 647-42-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 647-42-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Featured ProductsMultiple SpecificationsSpot supply
Product Name: 1H,1H,2H,2H-PERFLUORO-1-OCTANOL Request for quotationCAS: 647-42-7
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Spot supply
Product Name: 1H,1H,2H,2H-Perfluorooctanol Visit Supplier Webpage Request for quotationCAS: 647-42-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1H,1H,2H,2H-PERFLUORO-1-OCTANOL Request for quotation
CAS: 647-42-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 647-42-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History


