193353-34-3
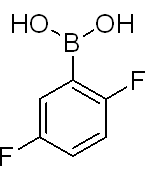
2,5-Difluorobenzeneboronic acid
CAS: 193353-34-3
Molecular Formula: C6H5BF2O2
193353-34-3 - Names and Identifiers
| Name | 2,5-Difluorobenzeneboronic acid |
| Synonyms | AKOS BRN-0301 RARECHEM AH PB 0165 2,5-Difluorophenylboronic acid 2,5-DIFLUOROPHENYLBORONIC ACID 2,5-FLUOROBENZENE BORONIC ACID 2,5-DIFLUOROBENZENEBORONIC ACID 2,5-Difluorobenzeneboronic acid 2,5-Difluorophenylboronic Acid (contains varying amounts of Anhydride) |
| CAS | 193353-34-3 |
| EINECS | 675-650-7 |
| InChI | InChI=1/C6H5FN2O2/c1-4-2-3-8-6(7)5(4)9(10)11/h2-3H,1H3 |
193353-34-3 - Physico-chemical Properties
| Molecular Formula | C6H5BF2O2 |
| Molar Mass | 157.91 |
| Density | 1.35±0.1 g/cm3(Predicted) |
| Melting Point | 105-110 °C (lit.) |
| Boling Point | 271.3±50.0 °C(Predicted) |
| Flash Point | 113.4°C |
| Vapor Presure | 0.0163mmHg at 25°C |
| Appearance | Crystalline powder |
| Color | White to light yellow |
| BRN | 8833254 |
| pKa | 7.29±0.58(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.533 |
| MDL | MFCD01863171 |
193353-34-3 - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R22 - Harmful if swallowed |
| Safety Description | S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37 - Wear suitable protective clothing and gloves. |
| WGK Germany | 3 |
| HS Code | 29319090 |
| Hazard Class | IRRITANT |
193353-34-3 - Reference Information
| properties | 2,5-difluorophenylboronic acid is white or light yellow solid at normal temperature and pressure. |
| use | 2,5-difluorophenylboronic acid can be used as an intermediate in pharmaceutical chemistry and organic synthesis. In the synthesis and transformation, the boric acid unit in the structure can be coupled to an aryl or alkyl group by Suzuki; boric acid can also be oxidized to hydroxyl to obtain the corresponding phenol compound; in addition, further derivatization reactions can be carried out through the coupling reaction of carbon and nitrogen with boric acid or carbon-oxygen bonds. |
| Preparation method | In argon environment, 1-bromo-2, 5-difluorobenzene and triisopropyl borate are cooled to -90 degrees in a dry tetrahydrofuran solution, and the reaction system is frozen for 30 minutes, then slowly add n-butyllithium 2.5M dropwise to the reaction mixture in hexane), and dilute the reaction mixture with cold tetrahydrofuran (3 ml). Stir the reaction mixture at -90 degrees for 1 hour, then raise the temperature of the reaction mixture to -10 degrees for reaction, and add aqueous hydrochloric acid to the reaction mixture after the reaction is over. The two phases are separated by a separatory funnel, and the water phase is extracted three times with ether, and the organic layers are combined. The obtained ether layer is dried with anhydrous sodium sulfate, filtered to remove the solid precipitate of sodium sulfate, and the obtained filtrate is concentrated under reduced pressure. Collect the remaining precipitate through filtration, and clean the precipitate with water (2 X 3 mL) and n-hexane (3x5 mL), the target product 2, 5-difluorophenylboronic acid can be obtained by drying in an environment of 50 degrees under vacuum for 8 hours. figure 2, synthesis route of 5-difluorophenylboronic acid |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2,5-Difluorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 193353-34-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2,5-Difluorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 193353-34-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,5-DIFLUOROPHENYLBORONIC ACID Request for quotation
CAS: 193353-34-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 193353-34-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2,5-Difluorophenylboronic acid Visit Supplier Webpage Request for quotation
CAS: 193353-34-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 193353-34-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2,5-Difluorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 193353-34-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2,5-Difluorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 193353-34-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,5-DIFLUOROPHENYLBORONIC ACID Request for quotation
CAS: 193353-34-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 193353-34-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2,5-Difluorophenylboronic acid Visit Supplier Webpage Request for quotation
CAS: 193353-34-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 193353-34-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



