189028-95-3
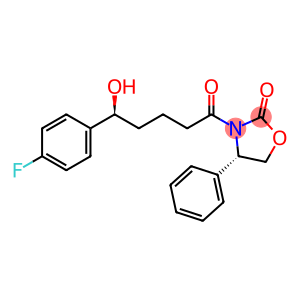
(4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE
CAS: 189028-95-3
Molecular Formula: C20H20FNO4
189028-95-3 - Names and Identifiers
189028-95-3 - Physico-chemical Properties
| Molecular Formula | C20H20FNO4 |
| Molar Mass | 357.38 |
| Density | 1.297 |
| Boling Point | 572.7±50.0 °C(Predicted) |
| Flash Point | 300.174°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 14.17±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.588 |
189028-95-3 - Reference Information
| overview | ezetimibe (Ezetimibe) is a drug jointly developed by Schering-Plough and Merck to treat hyperlipidemia. it was approved by FDA in 2002 under the trade name Zetia. Its chemical name is: 1-(4-fluorophenyl)-(3R)-[3-(4-fluorophenyl)-(3S)-hydroxypropyl]-(4S)-(4-hydroxyphenyl)-2-propionamide. Hyperlipidemia is an important risk factor for cardiovascular diseases such as atherosclerosis. Reducing the level of total cholesterol in plasma can reduce the risk of cardiovascular diseases. |
| Application | (4S)-3-[(5S)-5-(4-fluorophenyl)-5-hydroxyvalanoyl]-4-Phenyl -1, 3-oxazepentan-2-one is used as an intermediate for the preparation of ezetimibe. Ezetimibe's current clinical drugs for the treatment of hyperlipidemia mainly include cholesterol synthesis inhibitors, phenoxy acids and bile acid chelating agents. By acting on the brush border of small intestinal cells, ezetimibe can selectively inhibit the absorption of cholesterol in the biliary tract and food without affecting the absorption of fat-soluble vitamins, triglycerides and bile acids. |
| use | ezetimibe intermediate |
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: (4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE Visit Supplier Webpage Request for quotationCAS: 189028-95-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Ezetimibe Impurity 51 Visit Supplier Webpage Request for quotationCAS: 189028-95-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: (4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE Request for quotation
CAS: 189028-95-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 189028-95-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: (4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE Visit Supplier Webpage Request for quotation
CAS: 189028-95-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 189028-95-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE Visit Supplier Webpage Request for quotationCAS: 189028-95-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Ezetimibe Impurity 51 Visit Supplier Webpage Request for quotationCAS: 189028-95-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: (4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE Request for quotation
CAS: 189028-95-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 189028-95-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: (4S)-3-[(5R)-5-(4-FLUOROPHENYL)-5-HYDROXYPENTANOYL]-4-PHENYL-1,3-OXAZOLIDIN-2-ONE Visit Supplier Webpage Request for quotation
CAS: 189028-95-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 189028-95-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



