17557-76-5
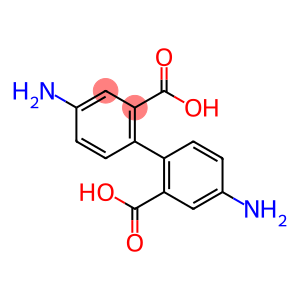
4,4'-Diaminobiphenyl-2,2'-Dicarboxylic acid
CAS: 17557-76-5
Molecular Formula: C14H12N2O4
17557-76-5 - Names and Identifiers
| Name | 4,4'-Diaminobiphenyl-2,2'-Dicarboxylic acid |
| Synonyms | DK1348 2,2'-DCB 2,2'-Dicarboxybenzidine 4,4'-Diamino-2,2'-biphenyldicarboxylic Acid 4,4'-DIAMINOBIPHENYL-2,2'-DICARBOXYLIC ACID 4,4'-Diaminobiphenyl-2,2'-Dicarboxylic acid 2,2'-Dicarboxylic acid-4,4'-Diaminobiphenyl 4,4'-Diamino-[1,1'-biphenyl]-2,2'-dicarboxylic acid [1,1'-Biphenyl]-2,2'-dicarboxylicacid, 4,4'-diaMino- |
| CAS | 17557-76-5 |
| EINECS | 1533716-785-6 |
| InChI | InChI=1/C14H12N2O4/c15-7-1-3-9(11(5-7)13(17)18)10-4-2-8(16)6-12(10)14(19)20/h1-6H,15-16H2,(H,17,18)(H,19,20) |
17557-76-5 - Physico-chemical Properties
| Molecular Formula | C14H12N2O4 |
| Molar Mass | 272.26 |
| Density | 1.476±0.06 g/cm3(Predicted) |
| Melting Point | 282 °C |
| Boling Point | 515.1±50.0 °C(Predicted) |
| Flash Point | 265.3°C |
| Vapor Presure | 1.96E-11mmHg at 25°C |
| pKa | 2.93±0.36(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.724 |
17557-76-5 - Introduction
4,4 '-Diaminobiphenyl-2,2'-Dicarboxylic acid, also known as terethalic acid, is an organic compound. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: Colorless crystalline solid
-Molecular formula: C8H6O4
-Molecular weight: 166.13g/mol
-Solubility: Slightly soluble in water, but better soluble in organic solvents such as alcohols, ethers and ketones.
-Melting Point: 300-305 ℃
-Stability: Stable at room temperature, but will decompose in the presence of high temperature, light and oxidants.
Use:
-Polyester fiber manufacturing: terethalic acid is an important raw material for polyterephthalic acid, which is used to manufacture polyester fibers, such as polyester threads, polyester fabrics, etc.
-Polyester resin production: Terephthalic acid reacts with ethylene glycol to generate polyester resin, which is used in the manufacture of plastic bottles, plastic films, etc.
-Dye synthesis: Terephthalic acid can be used to synthesize some organic dyes, widely used in textile, printing and other industries.
Method:
-Industrially, terephalic acid is mainly produced by the oxidation of Terephthalic acid. First, terephthalic acid is reacted with oxygen or an oxidizing agent to form an isophthaloquinone derivative. Then, the isophthaloquinone derivative was reduced with water to give Terephthalic acid.
-in the laboratory, para-diiodobenzoic acid can be obtained by iodination reaction of benzoic acid, and then 4,4 '-Diaminobiphenyl-2,2'-Dicarboxylic acid can be generated by deiodination reaction.
Safety Information:
- Terephthalic acid is generally considered to be a low-toxic and safe substance, without obvious irritation and carcinogenicity.
-but should avoid prolonged contact with the skin and inhalation of its dust.
-During the operation, care should be taken to avoid contact with the eyes and esophagus.
-When handling terethalic acid, it is recommended to wear a protective mask, gloves and safety glasses.
-During storage and transportation, keep it dry and away from fire and oxidizing agents.
Nature:
-Appearance: Colorless crystalline solid
-Molecular formula: C8H6O4
-Molecular weight: 166.13g/mol
-Solubility: Slightly soluble in water, but better soluble in organic solvents such as alcohols, ethers and ketones.
-Melting Point: 300-305 ℃
-Stability: Stable at room temperature, but will decompose in the presence of high temperature, light and oxidants.
Use:
-Polyester fiber manufacturing: terethalic acid is an important raw material for polyterephthalic acid, which is used to manufacture polyester fibers, such as polyester threads, polyester fabrics, etc.
-Polyester resin production: Terephthalic acid reacts with ethylene glycol to generate polyester resin, which is used in the manufacture of plastic bottles, plastic films, etc.
-Dye synthesis: Terephthalic acid can be used to synthesize some organic dyes, widely used in textile, printing and other industries.
Method:
-Industrially, terephalic acid is mainly produced by the oxidation of Terephthalic acid. First, terephthalic acid is reacted with oxygen or an oxidizing agent to form an isophthaloquinone derivative. Then, the isophthaloquinone derivative was reduced with water to give Terephthalic acid.
-in the laboratory, para-diiodobenzoic acid can be obtained by iodination reaction of benzoic acid, and then 4,4 '-Diaminobiphenyl-2,2'-Dicarboxylic acid can be generated by deiodination reaction.
Safety Information:
- Terephthalic acid is generally considered to be a low-toxic and safe substance, without obvious irritation and carcinogenicity.
-but should avoid prolonged contact with the skin and inhalation of its dust.
-During the operation, care should be taken to avoid contact with the eyes and esophagus.
-When handling terethalic acid, it is recommended to wear a protective mask, gloves and safety glasses.
-During storage and transportation, keep it dry and away from fire and oxidizing agents.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 4,4'-Diamino-[1,1'-biphenyl]-2,2'-dicarboxylic acid Visit Supplier Webpage Request for quotationCAS: 17557-76-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 4,4'-Diamino-[1,1'-biphenyl]-2,2'- dicarboxylic acid Request for quotationCAS: 17557-76-5
Tel: +86-022-83726202
Email: sales5@chinatechem.com
Mobile: +8618222270857 +8618222265956
QQ: 2081145741
Wechat: 18222270857 18222265956
Product Name: 4,4'-DIAMINOBIPHENYL-2,2'-DICARBOXYLIC ACID Request for quotation
CAS: 17557-76-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 17557-76-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 4,4'-DIAMINOBIPHENYL-2,2'-DICARBOXYLIC ACID Visit Supplier Webpage Request for quotation
CAS: 17557-76-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 17557-76-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4,4'-Diamino-[1,1'-biphenyl]-2,2'-dicarboxylic acid Visit Supplier Webpage Request for quotationCAS: 17557-76-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 4,4'-Diamino-[1,1'-biphenyl]-2,2'- dicarboxylic acid Request for quotationCAS: 17557-76-5
Tel: +86-022-83726202
Email: sales5@chinatechem.com
Mobile: +8618222270857 +8618222265956
QQ: 2081145741
Wechat: 18222270857 18222265956
Product Name: 4,4'-DIAMINOBIPHENYL-2,2'-DICARBOXYLIC ACID Request for quotation
CAS: 17557-76-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 17557-76-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 4,4'-DIAMINOBIPHENYL-2,2'-DICARBOXYLIC ACID Visit Supplier Webpage Request for quotation
CAS: 17557-76-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 17557-76-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



