1743-86-8
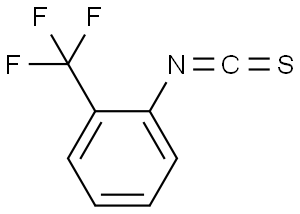
2-(TRIFLUOROMETHYL)PHENYL ISOTHIOCYANATE
CAS: 1743-86-8
Molecular Formula: C8H4F3NS
1743-86-8 - Names and Identifiers
1743-86-8 - Physico-chemical Properties
| Molecular Formula | C8H4F3NS |
| Molar Mass | 203.18 |
| Density | 1.346g/mLat 25°C(lit.) |
| Boling Point | 220°C(lit.) |
| Flash Point | 221°F |
| Specific Gravity | 1.346 |
| BRN | 2693552 |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Sensitive | Moisture Sensitive |
| Refractive Index | n20/D 1.566(lit.) |
| MDL | MFCD00039644 |
1743-86-8 - Risk and Safety
| Risk Codes | 34 - Causes burns |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S27 - Take off immediately all contaminated clothing. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | 2810 |
| WGK Germany | 3 |
| HS Code | 29309090 |
| Hazard Note | Harmful/Irritant |
| Hazard Class | 6.1 |
| Packing Group | III |
1743-86-8 - Nature
| boiling point | 220 °C(lit.) |
| density | 1.346 g/mL at 25 °C(lit.) |
| refractive index | n20/D 1.566(lit.) |
| flash point | 221 °F |
| storage conditions | Inert atmosphere,2-8°C |
| Specific gravity | 1.346 |
| sensitivity | Moisture Sensitive |
| BRN | 2693552 |
Last Update:2024-04-10 22:29:15
1743-86-8 - Reference Information
| Introduction | 2-(trifluoromethyl) phenylisothiocyanate is an organic intermediate that can be used as a raw material to prepare 2-trifluoromethylphenyldithiocarbate triethylammonium salt, and then it is substituted with ditribromine 1, the reaction of 3-dipyridinium hydrochloride propane gives 2-(trifluoromethyl) phenylisothiocyanate. |
| Application | 2-(trifluoromethyl) phenylisothiocyanate can be used to prepare a class of anti-inflammatory activity with the following structure Thiethylethyl urea/thiourea dimer small molecule compounds. These compounds have anti-inflammatory activity and improve the survival rate of septic shock mice. |
| preparation | 1) add 30.0 mmol2-trifluoromethylaniline and 10.0mmol triethylenediamine into a 50mL eggplant-shaped bottle, add 15.0mL toluene, start stirring at room temperature, add 1.80 mLCS2 dropwise at the same time, then stir at room temperature for 10 hours, filter, the solid obtained by suction filtration is naturally dried to obtain yellow powder 2-trifluoromethylphenyldithiocarboxylic acid triethylammonium salt (1.32g,12.6%). 2) Add 3.88 mmol2-trifluoromethylphenyl isothiocyanate to a 50mL eggplant-shaped flask, add 16.0mL acetonitrile and 7.90mmol triethylamine in turn, stir at 0 ℃, add 2.00mmol of ditribromo-1, 3-dipyridinium hydrochloride propane in batches during the stirring process, keep the temperature at 0 ℃ for 30min after adding, then stir at room temperature for reaction, and use TLC (thin layer chromatography, developing agent: petroleum ether) detection reaction is complete. When the reaction is 16 hours, TLC detection finds that the reaction is complete, and then the filter cake is filtered, the resulting filter cake is recovered, the resulting filtrate is concentrated, and the concentrated filtrate is subjected to column chromatography to obtain yellow oil-like 2-(trifluoromethyl) phenylisothiocyanate (0.063g,7.75%). IR(KBr):2028(νN = C = S)cm-1,1579cm-1,1471[νC = C (benzene ring carbon frame)]cm-1. |
Last Update:2024-04-10 22:29:15
1743-86-8 - Security information
| dangerous goods mark | C,Xi,T |
| hazard category code | 34 |
| safety instructions | 26-27-36/37/39-45 |
| dangerous goods transport number | 2810 |
| WGK Germany | 3 |
| Hazard Note | Harmful/Irritant |
| HazardClass | 6.1 |
| PackingGroup | III |
| customs code | 29309090 |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2-(Trifluoromethyl)phenyl isothiocyanate Visit Supplier Webpage Request for quotationCAS: 1743-86-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Alpha,Alpha,Alpha-Trifluoro-o-tolyl isocyanate Request for quotation
CAS: 1743-86-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1743-86-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-(TRIFLUOROMETHYL)PHENYL ISOTHIOCYANATE Request for quotation
CAS: 1743-86-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1743-86-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2-(Trifluoromethyl)Phenyl Isothiocyanate Visit Supplier Webpage Request for quotation
CAS: 1743-86-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1743-86-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple Specifications
Product Name: 2-(TRIFLUOROMETHYL)PHENYLISOTHIOCYANATE Request for quotationCAS: 1743-86-8
Tel: +86 021-37285211
Email: info@yudiaochem.com
Mobile: +86 18964703211
QQ: 122752498
Wechat: yudiaochem
Spot supply
Product Name: 2-(Trifluoromethyl)phenyl isothiocyanate Visit Supplier Webpage Request for quotationCAS: 1743-86-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Alpha,Alpha,Alpha-Trifluoro-o-tolyl isocyanate Request for quotation
CAS: 1743-86-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1743-86-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-(TRIFLUOROMETHYL)PHENYL ISOTHIOCYANATE Request for quotation
CAS: 1743-86-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1743-86-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2-(Trifluoromethyl)Phenyl Isothiocyanate Visit Supplier Webpage Request for quotation
CAS: 1743-86-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1743-86-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple Specifications
Product Name: 2-(TRIFLUOROMETHYL)PHENYLISOTHIOCYANATE Request for quotationCAS: 1743-86-8
Tel: +86 021-37285211
Email: info@yudiaochem.com
Mobile: +86 18964703211
QQ: 122752498
Wechat: yudiaochem
View History
1743-86-8
6-Oxabicyclo[3.1.0]hexan-2-ol,4-(methoxymethoxy)-,(1-alpha-,2-bta-,4-alpha-,5-alpha-)-(9CI)
6-Oxabicyclo[3.1.0]hexan-2-ol,4-(methoxymethoxy)-,(1-alpha-,2-bta-,4-alpha-,5-alpha-)-(9CI)




