172319-75-4
Thiophene, 2-bromo-5-propyl-
CAS: 172319-75-4
Molecular Formula: C7H9BrS
172319-75-4 - Names and Identifiers
| Name | Thiophene, 2-bromo-5-propyl- |
| Synonyms | 5-Bromo-2-Propylthiophene 2-Bromo-5-Propylthiophene 2-bromo-5-propylthyophene 2-broMo-5-propylthyophene Thiophene, 2-bromo-5-propyl- |
| CAS | 172319-75-4 |
172319-75-4 - Physico-chemical Properties
| Molecular Formula | C7H9BrS |
| Molar Mass | 205.12 |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
172319-75-4 - Reference Information
| Overview | the synthesis of thiophene polymers can be divided into two categories: Chemical oxidation polymerization and electrochemical polymerization. During chemical polymerization, the physical and chemical properties of the polymer will be affected by many factors such as the concentration of the monomer, the nature of the oxidant or reducing agent, the polymerization temperature, the polymerization atmosphere, the nature of the mixing agent and the doping degree. |
| Applications | sensors, polymer light-emitting diodes (PLEDs) and other fields. These electroactive and photoactive polymers are generally polymers based on units of aromatic rings such as thiophene, pyrrole, benzene, carbazole or the like, aromatic heterocycles or the like. In a large number of electrochromic materials, 2-bromo-5-n-propyl thiophene due to its high electronic conductivity and good redox properties, as well as in the visible and infrared region, fast response time, the remarkable stability and high contrast ratio make it an important class of electrochromic conjugated polymers. More importantly, 2-bromo-5-n-propylthiophene possesses easy band gap adjustability by modifying the polymer chain structure, and can exhibit different electrochromic properties. |
| preparation method | The synthesis methods of 2-bromo-5-n-propyl thiophene can be divided into two categories: chemical method and electrochemical method. The advantages and disadvantages of the two methods are also applicable to the synthesis of polythiophene and its derivatives. One of the most commonly used methods is the chemical synthesis of FeCl oxidation method, the synthesis of the vast majority of polythiophene and its derivatives have a strong applicability, in which the Fe "is both an oxidant and dopant, this allows the synthesis of the polymer to be facilitated. The advantage of using FeCl oxidation method to synthesize polythiophene is that it can be applied to large-scale production, but there are disadvantages such as relatively complex process, difficult to make thin film, relatively large film thickness and difficult to control. |
Last Update:2024-04-09 20:49:11
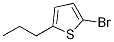
Supplier List
Spot supply
Product Name: 2-Bromo-5-propylthiophene Visit Supplier Webpage Request for quotationCAS: 172319-75-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Bromo-2-Propylthiophene Request for quotation
CAS: 172319-75-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 172319-75-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 5-Bromo-2-Propylthiophene Visit Supplier Webpage Request for quotation
CAS: 172319-75-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 172319-75-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Bromo-5-propylthiophene Visit Supplier Webpage Request for quotationCAS: 172319-75-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Bromo-2-Propylthiophene Request for quotation
CAS: 172319-75-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 172319-75-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 5-Bromo-2-Propylthiophene Visit Supplier Webpage Request for quotation
CAS: 172319-75-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 172319-75-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


