166943-39-1
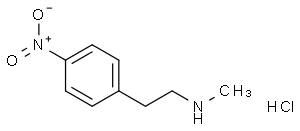
N-methyl-2-(4-nitrophenyl)ethylamine hydrochloride
CAS: 166943-39-1
Molecular Formula: C9H13ClN2O2
166943-39-1 - Names and Identifiers
| Name | N-methyl-2-(4-nitrophenyl)ethylamine hydrochloride |
| Synonyms | AURORA KA-7763 N-Methyl-4-NitrophenylethylaMine HCl METHYL-[2-(4-NITRO-PHENYL)-ETHYL]-AMINE N-METHYL-[2-(4-NITRO-PHENYL)-ETHYL]-AMINE N-METHYL-4-NITROPHENETHYLAMINE HYDROCHLORIDE N-Methyl-4-nitrophenethylamine hydrochloride N-methyl-2-(4-nitrophenyl)ethylamine hydrochloride N-METHYL-2-(4-NITROPHENYL)ETHYLAMINE HYDROCHLORIDE N-Methyl-2-(4-nitrophenyl)ethanaMine hydrochloride METHYL-[2-(4-NITRO-PHENYL)-ETHYL]-AMINE HYDROCHLORIDE |
| CAS | 166943-39-1 |
| EINECS | 1592732-453-0 |
| InChI | InChI=1/C9H12N2O2.ClH/c1-10-7-6-8-2-4-9(5-3-8)11(12)13;/h2-5,10H,6-7H2,1H3;1H |
| InChIKey | VGJDSNZOPPZCTB-UHFFFAOYSA-N |
166943-39-1 - Physico-chemical Properties
| Molecular Formula | C9H13ClN2O2 |
| Molar Mass | 216.66 |
| Melting Point | 222-227°C |
| Boling Point | 335.8°C at 760 mmHg |
| Flash Point | 156.9°C |
| Solubility | DMSO, Methanol |
| Vapor Presure | 8.36E-05mmHg at 25°C |
| Appearance | Solid |
| Color | White to Light Yellow |
| Storage Condition | Sealed in dry,Room Temperature |
166943-39-1 - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
166943-39-1 - Reference Information
| Application | N-methyl-4-nitrophenylethylamine hydrochloride is a pharmaceutical intermediate, which can be used to synthesize other compounds with certain biological activity Such as dofetilide. Dofetilide is an oral antiarrhythmic drug for atrial fibrillation developed by Pfizer in the United States. It has a high reversal of sinus rhythm. It was first listed in the United States in May 2000 and is a Class III antiarrhythmic drug. The anti-arrhythmic dofetilide is disclosed in the European patent EP-A-0245997. Its patents of the same family include US4959366 (sub-case application US5079248), CN1014529, etc. The disclosed patent describes a variety of synthesis methods of dofetilide, but they all have the problem of difficult raw materials and high industrial production costs. Dofetilide is a methanesulfonamide derivative structurally similar to the antiarrhythmic drug sotalol. Its pharmacological mechanism is to exert the effect of class III antiarrhythmic drugs by inhibiting rapid potassium current (Ikr) and increasing action potential duration (QT interval). It was approved by the FDA of the U.S. Food and Drug Administration in 1999 and was first listed in the United States in May 2000. A large number of clinical studies have shown that dofetilide can be used to treat and prevent atrial rhythm disorders and paroxysmal supraventricular tachycardia, and is better than other antiarrhythmic drugs for the conversion of newly occurring atrial fibrillation and atrial flutter. It can be used to prevent the occurrence of ventricular tachycardia and relieve the condition of patients with heart failure. It has the advantages of significant curative effect and relatively small adverse reactions. It is an anti-arrhythmia drug with strong effect and good selectivity. N-methyl -4-nitrophenylethylamine hydrochloride is an important intermediate in the synthesis of dofetilide. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: N-methyl-2-(4-nitrophenyl)ethylamine hydrochloride Visit Supplier Webpage Request for quotationCAS: 166943-39-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-Methyl-4-nitrophenethylamine hydrochloride Request for quotation
CAS: 166943-39-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 166943-39-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: N-Methyl-4-Nitrophenethylamine Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 166943-39-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 166943-39-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: N-methyl-2-(4-nitrophenyl)ethylamine hydrochloride Visit Supplier Webpage Request for quotationCAS: 166943-39-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-Methyl-4-nitrophenethylamine hydrochloride Request for quotation
CAS: 166943-39-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 166943-39-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: N-Methyl-4-Nitrophenethylamine Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 166943-39-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 166943-39-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


