16673-34-0
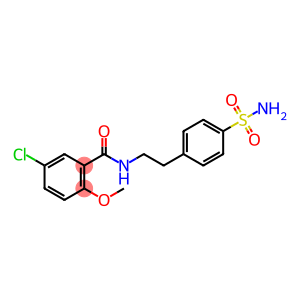
4-[2-(2-Methoxy-5-chlorobenzamido)ethyl]benzene sulfonamide
CAS: 16673-34-0
Molecular Formula: C16H17ClN2O4S
16673-34-0 - Names and Identifiers
16673-34-0 - Physico-chemical Properties
| Molecular Formula | C16H17ClN2O4S |
| Molar Mass | 368.84 |
| Density | 1.356±0.06 g/cm3(Predicted) |
| Melting Point | 209-214 °C |
| Solubility | Soluble in DMSO (up to 45 mg/ml). |
| Appearance | neat |
| Color | White |
| pKa | 10.14±0.10(Predicted) |
| Storage Condition | 2-8°C |
| Stability | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 3 months. |
| Refractive Index | 1.597 |
| Physical and Chemical Properties | Crystalline compound. Melting point 185-200 °c. |
16673-34-0 - Risk and Safety
| Hazard Symbols | T - Toxic |
| Risk Codes | R23 - Toxic by inhalation R24 - Toxic in contact with skin R25 - Toxic if swallowed |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 1 |
| HS Code | 2935909550 |
16673-34-0 - Reference Information
| Application | sulfonylurea is one of the earliest oral hypoglycemic drugs used in clinic, is the second generation of sulfonylurea hypoglycemic drugs. Its hypoglycemic effect is strong, low price, easy to use, in the treatment of diabetes, especially in the primary hospital and the majority of rural areas is still widely used. According to the 2012 diabetes treatment drug market research report, sulfonylureas accounted for 33 of the total hypoglycemic drug market. Among them, glibenclamide, glipizide, gliquidone and other sulfonylurea hypoglycemic drugs into the diabetes treatment drug clinical drug market share in the top 15. 4-[2-(5-chloro-2-methoxybenzoylamino) ethyl] benzenesulfonamide is an impurity produced during the synthesis of glibenclamide. |
| preparation | 4-[2-(5-chloro-2-methoxybenzoylamino) the preparation of ethyl] benzenesulfonamide can use salicylic acid as the starting material, salicylic acid is chlorinated with chlorine under light to generate 5-chlorosalicylic acid, and then methylated with dimethyl sulfate to obtain 5-chloro-2-methoxybenzoic acid, the latter was chlorinated with thionyl chloride to give 5-chloro-2-methoxybenzoyl chloride, which was then condensed with aniline to give n-phenethyl-5-chloro-2-methoxybenzamide, further, chlorosulfonation with chlorosulfonic acid and amination with ammonia water were used to prepare the product. 2-methoxy-5-chloro-benzoic acid can also be used as a raw material, first react with thionyl chloride and methanol to produce 2-methoxy-5-chloro-benzoic acid methyl ester, and then condense with phenylethylamine, the obtained amide was subjected to chlorosulfonation and hydrazine hydrolysis to obtain the target product 4-[2-(5-chloro-2-methoxybenzoylamino) ethyl] benzenesulfonamide. The preparation scheme is as follows: Figure 1 4-[2-(5-chloro-2-methoxybenzoylamino) ethyl] benzenesulfonamide preparation scheme |
| biological activity | NLRP3 inflam symptom Inhibitor I is an intermediate substrate for the synthesis of glibenclamide, which is an Inhibitor of NLRP3 inflam symptom. |
| Target | Value |
| Animal Model: | Experimental acute myocardial infarction (AMI) model in mice. |
| Dosage: | 100 mg/kg. |
| Administration: | Intraperitoneal administration 30 minutes prior to surgery, then every 6 hours for 3 additional doses. |
| Result: | Led to a significant >90% reduction in caspase-1 activity (reflective of the formation of an active inflammasome) in the heart tissue measured 24 hours after ischemia. Led to a significant reduction in the infarct size measured with TTC (>40% reduction) or troponin I levels (>70% reduction) when compared with vehicle alone. |
| Use | an intermediate of glibenclamide. |
| production method | 5-chlorosalicylic acid is generated by chlorination of chlorine with salicylic acid under light, then 5-chloro -2-methoxy benzoic acid was obtained by methylation with dimethyl sulfate, and the latter was chlorinated with thionyl chloride to obtain 5-chloro -2-methoxy benzoyl chloride, which was then condensed with aniline, the product was n-phenethyl-5-chloro-2-methoxybenzamide, which was further obtained by chlorosulfonic acid chlorosulfonation and ammonia amination. |
Last Update:2024-04-09 21:21:28
Supplier List
Spot supply
Product Name: 5-Chloro-2-methoxy-N-[2-(4-sulfamoylphenyl)ethyl]benzamide Visit Supplier Webpage Request for quotationCAS: 16673-34-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-(2-(5-Chloro-2-methoxybenzamido)ethyl)benzenesulfamide Request for quotation
CAS: 16673-34-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 16673-34-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: 5-Chloro-2-methoxy-N-[2-(4-sulfamoylphenyl)ethyl]benzamide Visit Supplier Webpage Request for quotationCAS: 16673-34-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-(2-(5-Chloro-2-methoxybenzamido)ethyl)benzenesulfamide Request for quotation
CAS: 16673-34-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 16673-34-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History

