1628471-64-6
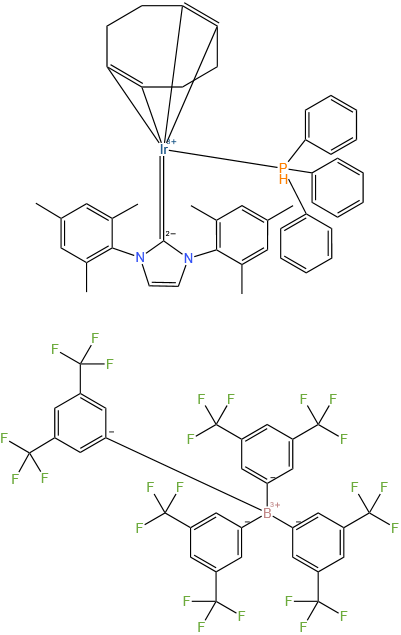
Triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene] iridium(I) tetrakis(3,5-bis(trifluoromethyl)phenylborate
CAS: 1628471-64-6
Molecular Formula: C79H60BF24IrN2P
1628471-64-6 - Names and Identifiers
1628471-64-6 - Physico-chemical Properties
| Molecular Formula | C79H60BF24IrN2P |
| Molar Mass | 1727.33 |
1628471-64-6 - Introduction
Triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene] iridium(I) tetrakis(3,5-bis(trifluoromethyl)phenylborate) is a complex complex with the chemical formula
[(COD)Ir(tpmd-IMes)][ B (C6F5)4]
Wherein, COD represents 1,5-cyclooctadiene, and tpmd-IMes represents 1,3-bis (2,4, 6-trimethylphenyl) imidazoline-2-methylene group. This complex has the following characteristics:
Nature:
1. The complex has high stability and catalytic activity;
2. It is blue or green in solution;
3. It is stable to air and water.
Use:
1. The complex can be used as a catalyst for catalytic hydrogenation and asymmetric catalysis in organic synthesis;
2. It can also be used for the synthesis and research of low oxidation state metals.
Preparation Method:
Triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene] iridium(I) tetrakis(3,5-bis(trifluoromethyl)phenylborate) can be prepared by the following steps:
1. Oxidation of Ir(III) to Ir(V) by a strong oxidant;
2. react with tpmd-IMes to generate (tpmd-IMes)Ir(V) compound;
3. generate (tpmd-IMes)Ir(COD) complex with COD;
4. react with B(C6F5)3 to form (tpmd-IMes)Ir(COD)(B(C6F5)3)3 complex;
5. by reacting with methylene triphenylphosphine to generate the final complex.
Safety Information:
The detailed safety information and hazards of this complex have not been extensively studied. When working with such organometallic compounds, good laboratory practices should be followed and appropriate engineering controls and personal protective measures should be used. If necessary, operate in a well-ventilated environment and avoid contact with oxygen and water. It is recommended to operate in a specialized laboratory or an organometallic chemistry laboratory.
[(COD)Ir(tpmd-IMes)][ B (C6F5)4]
Wherein, COD represents 1,5-cyclooctadiene, and tpmd-IMes represents 1,3-bis (2,4, 6-trimethylphenyl) imidazoline-2-methylene group. This complex has the following characteristics:
Nature:
1. The complex has high stability and catalytic activity;
2. It is blue or green in solution;
3. It is stable to air and water.
Use:
1. The complex can be used as a catalyst for catalytic hydrogenation and asymmetric catalysis in organic synthesis;
2. It can also be used for the synthesis and research of low oxidation state metals.
Preparation Method:
Triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene] iridium(I) tetrakis(3,5-bis(trifluoromethyl)phenylborate) can be prepared by the following steps:
1. Oxidation of Ir(III) to Ir(V) by a strong oxidant;
2. react with tpmd-IMes to generate (tpmd-IMes)Ir(V) compound;
3. generate (tpmd-IMes)Ir(COD) complex with COD;
4. react with B(C6F5)3 to form (tpmd-IMes)Ir(COD)(B(C6F5)3)3 complex;
5. by reacting with methylene triphenylphosphine to generate the final complex.
Safety Information:
The detailed safety information and hazards of this complex have not been extensively studied. When working with such organometallic compounds, good laboratory practices should be followed and appropriate engineering controls and personal protective measures should be used. If necessary, operate in a well-ventilated environment and avoid contact with oxygen and water. It is recommended to operate in a specialized laboratory or an organometallic chemistry laboratory.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: Triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene] iridium(I) tetrakis(3,5-bis(trifluoromethyl)phenylborate Request for quotation
CAS: 1628471-64-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1628471-64-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene] iridium(I) tetrakis(3,5-bis(trifluoromethyl)phenylborate Request for quotation
CAS: 1628471-64-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1628471-64-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
