161075-00-9
Hexafluoropropene, oxidized, oligomers, reduced, fluorinated
CAS: 161075-00-9
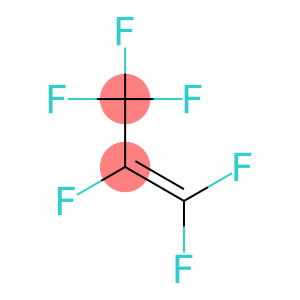
Molecular Formula: C3F6
161075-00-9 - Names and Identifiers
| Name | Hexafluoropropene, oxidized, oligomers, reduced, fluorinated |
| Synonyms | Hexafluoropropene, oxidized, oligomers, reduced, fluorinated |
| CAS | 161075-00-9 |
| EINECS | 500-537-5 |
161075-00-9 - Physico-chemical Properties
| Molecular Formula | C3F6 |
| Molar Mass | 150.0225192 |
| Density | 1.676[at 20℃] |
| Boling Point | 62℃[at 101 325 Pa] |
| Water Solubility | 470μg/L at 20℃ |
| Vapor Presure | 19.25kPa at 20℃ |
161075-00-9 - Introduction
Hexafluopropene (Hexafluoropropene) is a highly fluorinated compound. It is a colorless gas with a low boiling point and a high combustion temperature.
About the properties of hexafluopropene:
1. Smell: hexafluopropene almost no smell.
2. Stability: It is relatively stable at room temperature, but it will decompose under fire conditions to produce toxic gases.
3. Solubility: hexafluopropene is almost insoluble in water, but can be dissolved in organic solvents.
About the use of hexafluopropene:
1. As a synthetic rubber and plastic additive to improve its chemical resistance.
2. Used for the manufacture of special materials, such as coatings, sealing materials and electronic components.
3. As a high temperature lubricant and heat transfer medium.
4. Used for cleaning and manufacturing processes in the semiconductor industry.
preparation method of hexafluopropene:
Hexafluoropropene is usually prepared by the reaction of trifluoromethane (CHF3) and trifluoromethane (F2) catalyzed by fluorine gas and aluminum trichloride (AlCl3) catalyst.
The preparation equation is as follows:
3 CHF3 AlCl3 → C3F6 3 HCl
Hexafluoropropene, oxidized, oligomers, reduced, fluorinated (oxidized Hexafluoropropene, oligomer, reduction, fluorination) is the oxidation, oligomerization, reduction, different compound forms obtained by treatment such as fluorination. These compounds can be used in specific areas of research and applications, for example as components of high-performance polymers, surface coatings and nanomaterials.
About the properties of hexafluopropene:
1. Smell: hexafluopropene almost no smell.
2. Stability: It is relatively stable at room temperature, but it will decompose under fire conditions to produce toxic gases.
3. Solubility: hexafluopropene is almost insoluble in water, but can be dissolved in organic solvents.
About the use of hexafluopropene:
1. As a synthetic rubber and plastic additive to improve its chemical resistance.
2. Used for the manufacture of special materials, such as coatings, sealing materials and electronic components.
3. As a high temperature lubricant and heat transfer medium.
4. Used for cleaning and manufacturing processes in the semiconductor industry.
preparation method of hexafluopropene:
Hexafluoropropene is usually prepared by the reaction of trifluoromethane (CHF3) and trifluoromethane (F2) catalyzed by fluorine gas and aluminum trichloride (AlCl3) catalyst.
The preparation equation is as follows:
3 CHF3 AlCl3 → C3F6 3 HCl
Hexafluoropropene, oxidized, oligomers, reduced, fluorinated (oxidized Hexafluoropropene, oligomer, reduction, fluorination) is the oxidation, oligomerization, reduction, different compound forms obtained by treatment such as fluorination. These compounds can be used in specific areas of research and applications, for example as components of high-performance polymers, surface coatings and nanomaterials.
Last Update:2024-04-09 21:54:55
Supplier List
Product Name: Hexafluoropropene, oxidized, oligomers, reduced, fluorinated Request for quotation
CAS: 161075-00-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 161075-00-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: Hexafluoropropene, oxidized, oligomers, reduced, fluorinated Request for quotation
CAS: 161075-00-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 161075-00-9
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
View History
161075-00-9
二乙腈亚胺
propyl dihydrogen phosphate
478249-76-2
1215561-37-7
7-HYDROXY-6-DODECANONE
Carbonic acid, chloromethyl 1-methylethyl ester
2-(4-chloro-2-(trifluoromethyl)phenyl)acetic acid
二乙腈亚胺
propyl dihydrogen phosphate
478249-76-2
1215561-37-7
7-HYDROXY-6-DODECANONE
Carbonic acid, chloromethyl 1-methylethyl ester
2-(4-chloro-2-(trifluoromethyl)phenyl)acetic acid
