15489-90-4
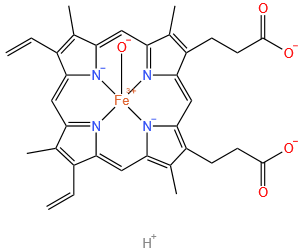
dihydrogen hydroxy[3,8,13,17-tetramethyl-7,12-divinyl-21H,23H-porphine-2,18-dipropionato(4-)-N21,N22,N23,N24]ferrate(2-)
CAS: 15489-90-4
Molecular Formula: C34H33FeN4O5
15489-90-4 - Names and Identifiers
15489-90-4 - Physico-chemical Properties
| Molecular Formula | C34H33FeN4O5 |
| Molar Mass | 633.49 |
| Melting Point | 180 °C |
| Boling Point | 1128.5°C at 760 mmHg |
| Flash Point | 636.3°C |
| Solubility | DMF (Slightly), DMSO (Slightly, Heated, Sonicated), Pyridine (Slightly, Heated, |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | solid |
| Color | Black |
| Merck | 14,4635 |
| Storage Condition | 2-8°C |
| Stability | Light Sensitive |
15489-90-4 - Risk and Safety
| WGK Germany | 3 |
| RTECS | NO6725000 |
| Toxicity | LD50 i.v. in rats: 4.32 mg/100 g (Lips) |
15489-90-4 - Reference Information
| introduction | heme iron, also known as heme, is a dark purple luster fine needle-like crystal or dark brown particles and powder. Slightly special smell. Very unstable and easy to oxidize. Insoluble in water. Used as an iron fortifier, its absorption rate is about 3 times higher than that of ordinary iron agents, and the iron content is 1.0% ~ 2.5%. |
| use | 1. heme iron is iron bound to porphyrin (porphyrin) in hemoglobin and myoglobin. it is directly absorbed by intestinal mucosal epithelial cells in the form of porphyrin iron. Generally, it is not affected by inhibitory factors such as phytate or promoting factors such as vitamin C. 2. Nutritional fortifier, blood tonic and food additive for iron. Determination of copper. Biochemical research. |
Last Update:2024-04-09 21:21:28
Supplier List
Spot supply
Product Name: Hematin porcine Visit Supplier Webpage Request for quotationCAS: 15489-90-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: HEMATIN Request for quotation
CAS: 15489-90-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 15489-90-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: Hematin Visit Supplier Webpage Request for quotationCAS: 15489-90-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Hematin porcine Visit Supplier Webpage Request for quotationCAS: 15489-90-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: HEMATIN Request for quotation
CAS: 15489-90-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 15489-90-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: Hematin Visit Supplier Webpage Request for quotationCAS: 15489-90-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


