15097-38-8
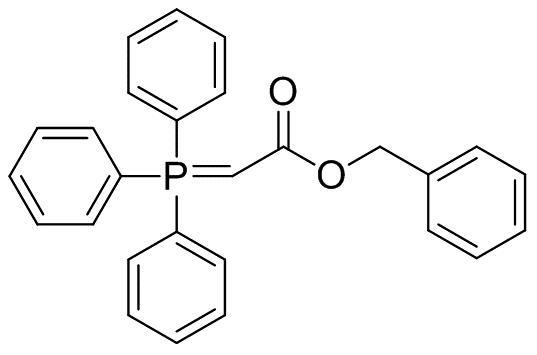
Benzyl (triphenylphosphoranylidene)acetate
CAS: 15097-38-8
Molecular Formula: C27H23O2P
15097-38-8 - Names and Identifiers
15097-38-8 - Physico-chemical Properties
| Molecular Formula | C27H23O2P |
| Molar Mass | 410.44 |
| Density | 1.19±0.1 g/cm3(Predicted) |
| Melting Point | 120-122°C(lit.) |
| Boling Point | 571.1±43.0 °C(Predicted) |
| Appearance | Shape Powder, color White to off-white |
| Color | White to off-white |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| MDL | MFCD00191787 |
| Use | Uses for the Wittig reaction of tributylphosphine as a medium; for the synthesis of 1,2-dioxane for anti-pyramidal activity; for the organic catalysis of phosphorus ylide and unsaturated ketones The Michael type reaction/Wittig reaction; used for the stereoselective phosphine-catalyzed cyclization addition to form spirocyclopentene indole; used for the enantioselective synthesis of pantothenic acid; used for the preparation of phosphorus ylide from orthophosphorus and acetic anhydride |
15097-38-8 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29319019 |
15097-38-8 - Introduction
(benzyloxycarbonylmethylene) triphenylphosphine is a phosphine ylide reagent, which can be used to prepare optically active α-aminopimelate L-2-aminopelic acid -1-ethyl ester.
Last Update:2024-04-10 22:29:15
15097-38-8 - Reference Information
| Introduction | (benzyloxycarbonylene methylene) Triphenylphosphine is a phosphine ylide reagent, which can be used to prepare optically active α-aminoheptanedioate L-2-aminoheptano-1-ethyl ester. |
| Use | Used in the tributylphosphine-mediated allene Wittig reaction; used in the synthesis of anti-pyramidal activity 1, 2-dioxane; used in the organocatalytic Michael reaction/Wittig reaction of phosphorus ylide and unsaturated ketones; used for stereoselective phosphine-catalyzed cyclization addition to form spirocyclopentene indole; used for enantioselective synthesis of pantothenic acid; used to prepare phosphorus ylide from n-phosphorus and acetic anhydride |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Benzyl (triphenylphosphoranylidene)acetate Visit Supplier Webpage Request for quotationCAS: 15097-38-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Benzyl(triphenylphosphoranylidene)acetate Visit Supplier Webpage Request for quotationCAS: 15097-38-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-Butyl-Aminopropyl Dimethoxy Methylsilane Request for quotation
CAS: 15097-38-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 15097-38-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Benzyl (triphenylphosphoranylidene)acetate Request for quotation
CAS: 15097-38-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 15097-38-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Benzyl (Triphenylphosphoranylidene)Acetate Visit Supplier Webpage Request for quotation
CAS: 15097-38-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 15097-38-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Benzyl (triphenylphosphoranylidene)acetate Visit Supplier Webpage Request for quotationCAS: 15097-38-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Benzyl(triphenylphosphoranylidene)acetate Visit Supplier Webpage Request for quotationCAS: 15097-38-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-Butyl-Aminopropyl Dimethoxy Methylsilane Request for quotation
CAS: 15097-38-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 15097-38-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Benzyl (triphenylphosphoranylidene)acetate Request for quotation
CAS: 15097-38-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 15097-38-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Benzyl (Triphenylphosphoranylidene)Acetate Visit Supplier Webpage Request for quotation
CAS: 15097-38-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 15097-38-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




