13765-19-0
calcium chromate
CAS: 13765-19-0
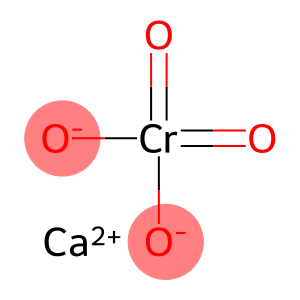
Molecular Formula: CaCrO4
13765-19-0 - Names and Identifiers
| Name | calcium chromate |
| Synonyms | C.I.77223 CalciumChromate CALCIUM CHROMATE calcium chromate calciumchromate(vi CalciumChromeYellow C.I.Pigment yellow 33 Calcium chromate (as Cr) |
| CAS | 13765-19-0 |
| EINECS | 237-366-8 |
| InChI | InChI=1/Ca.Cr.4O/q+2;;;;2*-1/rCa.CrO4/c;2-1(3,4)5/q+2;-2 |
13765-19-0 - Physico-chemical Properties
| Molecular Formula | CaCrO4 |
| Molar Mass | 156.0717 |
| Density | 2.500 |
| Water Solubility | Soluble in acid and water. Insoluble in alcohol. |
| Appearance | Powder |
| Color | Yellow |
| Exposure Limit | ACGIH: TWA 0.0002 mg/m3; STEL 0.0005 mg/m3 (Skin)OSHA: Ceiling 0.1 mg/m3NIOSH: IDLH 15 mg/m3; TWA 0.0002 mg/m3 |
| Solubility Product Constant(Ksp) | pKsp: 3.15 |
| Physical and Chemical Properties | The main component of calcium chrome yellow is calcium chromate (CaCrO4). The appearance was lemon yellow powder, the theoretical content of chromium trioxide was 64.1%, and the actual product also had a chromium trioxide content close to 60%. Since the atomic weight of calcium (Ca) is only 40.08, it is the highest content of chromium trioxide in chromate anti-rust pigment. The main features of water solubility is high, calcium chrome yellow water solubility is much higher than other varieties, plus high chromium trioxide content, so the toxicity of all chromate anti-rust pigment. |
| Use | Used as analytical reagent and oxidant inorganic anti-rust pigment. After the preparation of anti-rust paint, because it can provide sufficient chromate ions, the passivation effect is strengthened, and the anti-rust performance is improved. However, due to the solubility of water is too large, causing the decline of water resistance of the paint film, it is necessary to play its specialty, to choose the resin with good water resistance as the base material. Such as calcium chrome yellow calcium yellow perchloroethylene primer, calcium yellow chlorinated rubber primer, calcium yellow epoxy ester primer, etc. Some anti-rust primer formula also uses calcium chrome yellow, can also be used with zinc chrome yellow, chrome yellow, barium chrome yellow, etc., in order to obtain the effect of each other. |
13765-19-0 - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R45 - May cause cancer R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S53 - Avoid exposure - obtain special instructions before use. S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | 3288 |
| RTECS | GB2750000 |
| TSCA | Yes |
| Hazard Class | 5.1 |
| Packing Group | III |
13765-19-0 - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Use | inorganic anti-rust pigment. After the preparation of anti-rust paint, because it can provide sufficient chromate ions, the passivation effect is strengthened, and the anti-rust performance is improved. However, due to the solubility of water is too large, causing the decline of water resistance of the paint film, it is necessary to play its specialty, to choose the resin with good water resistance as the base material. Such as calcium chrome yellow calcium yellow perchloroethylene primer, calcium yellow chlorinated rubber primer, calcium yellow epoxy ester primer, etc. Some anti-rust primer formula also uses calcium chrome yellow, can also be used with zinc chrome yellow, chrome yellow, barium chrome yellow, etc., in order to obtain the effect of each other. used as analytical reagent and oxidant |
| production method | calcium carbonate method. Calcium carbonate is used as raw material to react calcium carbonate with chromic anhydride to produce calcium chromate and carbon dioxide. The reaction equation is as follows: CaCO3 CrO3 → CaCrO4 CO2 operation method: 30 parts of calcium carbonate, 31 parts of chromic anhydride and circulating mother liquor are prepared into feed solution and then sent to a steam heating reactor with stirring for reaction, heating to boiling reaction for more than 2H is generated in calcium chromate, reaction to the end, with a small amount of fine lime emulsion neutralization to pH 8, and then filtered, separated and dried to product. |
| category | oxidant |
| toxicity grade | high toxicity |
| Acute toxicity | oral-rat LD50: 327 mg/kg |
| explosive hazard characteristics | reaction with reductant |
| flammability hazard characteristics | open flame combustion with boron mixture |
| storage and transportation characteristics | The warehouse is ventilated and dried at low temperature; It is stored separately from organic substances and reducing agents |
| extinguishing agent | water, foam, carbon dioxide, dry powder |
| Occupational Standard | TWA 0.0001 mg (chromium)/m3 |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: CALCIUM CHROMATE Request for quotation
CAS: 13765-19-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 13765-19-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: CALCIUM CHROMATE Request for quotation
CAS: 13765-19-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 13765-19-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
