1365888-06-7
GDC-0810
CAS: 1365888-06-7
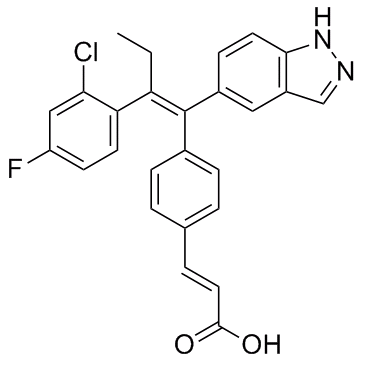
Molecular Formula: C26H20ClFN2O2
1365888-06-7 - Names and Identifiers
1365888-06-7 - Physico-chemical Properties
| Molecular Formula | C26H20ClFN2O2 |
| Molar Mass | 446.9 |
| Density | 1.342±0.06 g/cm3(Predicted) |
| Boling Point | 622.8±55.0 °C(Predicted) |
| Solubility | DMSO: ≥ 30 mg/mL |
| pKa | 13.94±0.40(Predicted) |
| Storage Condition | -20℃ |
| In vitro study | Overall, GDC-0810 bound ERα and ERβ with low nanomolar affinity in cell-free radioligand competition binding experiments. GDC-0810 had a weak inhibitory effect on CYP1A2, CYP2D6 or CYP3A4 (IC50>20 μm) and a moderate inhibitory effect on CYP2C9 and CYP2C19, with IC50 of 2.2 and 3.3 μm, respectively. It has a potent inhibitory effect on CYP2C8 with an IC50 of less than 0.1 μm. The weak effect of GDC-0810 on other nuclear hormone receptors shows that its selectivity is good. In transcriptional reporter analyses of mineralocorticoid, progesterone-A and glucocorticoid receptors, GDC-0810 had little activity against them with an IC50 greater than 1 μm. In binding experiments, GDC-0810 had little activity at the androgen receptor and the glucocorticoid receptor with an IC50 of greater than 4 μm and 0.99 μm, respectively. The GDC-0810 mediated reduction (depletion) of ERα is dependent on the 26S proteasome. GDC-0810 antagonized the ERα ligand-binding domain mutant in vitro and in vivo, and its effect on ER was tested in a cell-free E2 competitive binding assay. WT, ER.Y537S and ER. Binding to the D538G ligand-binding domain found that GDC-0810 could effectively displace E2 from the ligand-binding domain, although there was a slight increase in the IC50 for the mutant (WT: 2.6 nM vs. ER.Y537S: 5.5 nM and ER.D538G: 5.4 nM). GDC-0810 can compete with the co-activating molecule of PGC1α to dissociate it from the mutated ligand binding domain, indicating that GDC-0810 can cause a conformational change in mutant ER, the transition from an 'activate' conformation to an 'inactivate' conformation, albeit with a 5-to 7-fold decrease in biochemical activity compared to the wild-type ER. |
| In vivo study | Pharmacokinetic studies GDC-0810 found that GDC-0810 in various species belonging to the low clearance molecules, bioavailability is good, between 40%-60%. As a lipophilic carboxylic acid, it is highly binding (>99.5%) to plasma proteins with a low to moderate volume of distribution (Vss = 0.2 − 2.0 L/kg). GDC-0810 has good bioavailability among various species, and plays a strong and effective active role in the breast cancer transplantation tumor model sensitive to tamoxipen and resistant to tamoxipen. In in vitro and in vivo uterine models, GDC-0810 had mild estrogenic activity. |
1365888-06-7 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.238 ml | 11.188 ml | 22.376 ml |
| 5 mM | 0.448 ml | 2.238 ml | 4.475 ml |
| 10 mM | 0.224 ml | 1.119 ml | 2.238 ml |
| 5 mM | 0.045 ml | 0.224 ml | 0.448 ml |
Last Update:2024-01-02 23:10:35
1365888-06-7 - Reference Information
| biological activity | Brilanestrant (GDC-0810, ARN-81) is an effective ER-α binding agent (ER-α, IC50 = 6.1 nM; ER-β, IC50 = 8.8 nM), is its transcription antagonist, not activation. It has an effective inhibitory effect on the degradation of ER-α and MCF-7 cell activity. EC50 and IC50 are 0.7 nM and 2.5 nM respectively. Compared with other nuclear hormone receptors, it has good selectivity. |
| target | TargetValue ERα (Cell-free) 6.1 nM ERβ (Cell-free) 8.8 nM |
| Target | Value |
| ERα (Cell-free) | 6.1 nM |
| ERβ (Cell-free) | 8.8 nM |
| In vitro study | In the total experimental group of cell-free radioligand competition for binding, the GDC-0810 binds ERα and ERβ with low nanomolar affinity. GDC-0810 had a weak inhibitory effect on CYP1A2, CYP2D6 or CYP3A4 (IC50>20 μM) and a moderate inhibitory effect on CYP2C9 and CYP2C19, with IC50 of 2.2 and 3.3 μM, respectively. It has effective inhibitory effect on CYP2C8 with IC50 less than 0.1 μM. The weak effect of GDC-0810 on other nuclear hormone receptors indicates that its selectivity is good. In the transcriptional report analysis of mineralocorticoid, progesterone-A and glucocorticoid receptors, GDC-0810 have little activity on them, and IC50 is greater than 1 μM. In the binding experiment, GDC-0810 had little activity on androgen receptor and glucocorticoid receptor, IC50 was greater than 4 μM and 0.99 μM respectively. The reduction (consumption) of ERα mediated by GDC-0810 depends on 26S proteasome. GDC-0810 can antagonize ERα ligand binding region mutants in vivo and in vitro. in the cell-free E2 competitive binding experiment, it is found that GDC-0810 can effectively replace E2 from ligand binding region, although IC50 to mutant will slightly increase (WT: 2.6 nM vs. ER.Y537S: 5.5 nM and ER.D538G: 5.4 nM). GDC-0810 can compete with the co-activating molecule of PGC1α to dissociate it from the mutant ligand binding domain, indicating that GDC-0810 can change the conformational change of mutant ER from 'active' to 'inactive' conformation, although the biochemical activity is reduced by 5-7 times compared with that of wild type ER. |
| in vivo study | pharmacokinetic study of GDC-0810 found that GDC-0810 belongs to low clearance molecules in various species and has good bioavailability, ranging from 40% to 60%. As a lipophilic carboxylic acid, it can highly bind to plasma proteins (>99.5%) and has a low to medium distribution volume (Vss = 0.2 − 2.0 L/kg). GDC-0810 has good bioavailability among various species and plays a strong and effective active role in breast cancer transplanted tumor models sensitive to tamoxifen and tamoxifen resistance. In the in vivo and in vitro uterine model, GDC-0810 has mild estrogenic activity. |
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: Brilanestrant Visit Supplier Webpage Request for quotationCAS: 1365888-06-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: GDC-0810 Visit Supplier Webpage Request for quotationCAS: 1365888-06-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: GDC-0810 Request for quotation
CAS: 1365888-06-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1365888-06-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (E)-3-(4-((E)-2-(2-Chloro-4-fluorophenyl)-1-(1H-indazol-5-yl)but-1-en-1-yl)phenyl)acrylic acid Request for quotation
CAS: 1365888-06-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1365888-06-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: GDC-0810 Visit Supplier Webpage Request for quotation
CAS: 1365888-06-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1365888-06-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Brilanestrant Visit Supplier Webpage Request for quotationCAS: 1365888-06-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: GDC-0810 Visit Supplier Webpage Request for quotationCAS: 1365888-06-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: GDC-0810 Request for quotation
CAS: 1365888-06-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1365888-06-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (E)-3-(4-((E)-2-(2-Chloro-4-fluorophenyl)-1-(1H-indazol-5-yl)but-1-en-1-yl)phenyl)acrylic acid Request for quotation
CAS: 1365888-06-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1365888-06-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: GDC-0810 Visit Supplier Webpage Request for quotation
CAS: 1365888-06-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1365888-06-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




