1346601-82-8
Deferiprone-d3
CAS: 1346601-82-8
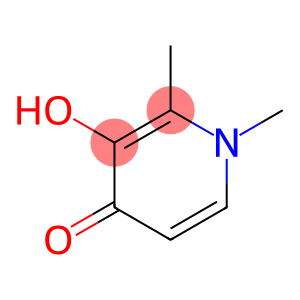
Molecular Formula: C7H9NO2
1346601-82-8 - Names and Identifiers
1346601-82-8 - Physico-chemical Properties
| Molecular Formula | C7H9NO2 |
| Molar Mass | 139.15 |
| Melting Point | >244°C (dec.) |
| Solubility | Methanol (Slightly, Heated), Water (Slightly, Heated) |
| Appearance | Solid |
| Color | White to Off-White |
| Storage Condition | -20°C Freezer |
1346601-82-8 - Introduction
Deferiprone-d3(Deferiprone-d3) is a deuterated deferriprone with properties similar to Deferiprone. It is a white solid and may exist in various crystal forms. The chemical formula of Deferiprone-d3 is C7H3D3N2O, and the relative molecular mass is 149.22g/mol.
Deferiprone-d3 is mainly used to treat blood diseases caused by iron overload, such as thalassemia or congenital superfluous iron storage disease. Its role is to help maintain a normal iron balance by binding and removing excess iron ions from the body. Deferiprone-d3 can form stable compounds with iron ions, making them easily eliminated from the body.
The preparation method of Deferiprone-d3 is generally synthesized by deuteration reaction. First, a deuteration reaction is performed by selecting an appropriate starting material to substitute a part or all of the hydrogen atoms. For example, in the preparation of Deferiprone, deuteration can be achieved by treating the precursor compound with sodium deuteride. In this way, Deferiprone-d3 can be obtained.
Regarding the safety information of the Deferiprone-d3, it is operated in accordance with the general chemical handling and storage methods. It should be stored in a dry, well-ventilated place, away from fire and flammable substances. During use, care should be taken to avoid inhalation, ingestion or contact with skin and eyes. If discomfort or accidental exposure occurs, you should immediately seek medical attention and bring the safety data sheet (SDS) of the compound.
It should be noted that the above information is for reference only. The specific use Deferiprone-d3 should follow the instructions and safety guidelines provided by the pharmaceutical company.
Deferiprone-d3 is mainly used to treat blood diseases caused by iron overload, such as thalassemia or congenital superfluous iron storage disease. Its role is to help maintain a normal iron balance by binding and removing excess iron ions from the body. Deferiprone-d3 can form stable compounds with iron ions, making them easily eliminated from the body.
The preparation method of Deferiprone-d3 is generally synthesized by deuteration reaction. First, a deuteration reaction is performed by selecting an appropriate starting material to substitute a part or all of the hydrogen atoms. For example, in the preparation of Deferiprone, deuteration can be achieved by treating the precursor compound with sodium deuteride. In this way, Deferiprone-d3 can be obtained.
Regarding the safety information of the Deferiprone-d3, it is operated in accordance with the general chemical handling and storage methods. It should be stored in a dry, well-ventilated place, away from fire and flammable substances. During use, care should be taken to avoid inhalation, ingestion or contact with skin and eyes. If discomfort or accidental exposure occurs, you should immediately seek medical attention and bring the safety data sheet (SDS) of the compound.
It should be noted that the above information is for reference only. The specific use Deferiprone-d3 should follow the instructions and safety guidelines provided by the pharmaceutical company.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: Deferiprone-d3 Visit Supplier Webpage Request for quotation
CAS: 1346601-82-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1346601-82-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Deferiprone-d3 Visit Supplier Webpage Request for quotation
CAS: 1346601-82-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1346601-82-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
