13451-18-8
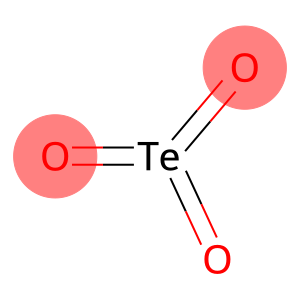
TELLURIUM TRIOXIDE
CAS: 13451-18-8
Molecular Formula: O3Te
13451-18-8 - Names and Identifiers
| Name | TELLURIUM TRIOXIDE |
| Synonyms | 13451-18-8 TELLURIUM OXIDE Tellurium(Ⅳ)oxide TELLURIUM TRIOXIDE Tellurium(VI) oxide Tellurium(VI) trioxide Tellurium oxide (119TeO2) |
| CAS | 13451-18-8 |
| EINECS | 236-620-5 |
13451-18-8 - Physico-chemical Properties
| Molecular Formula | O3Te |
| Molar Mass | 175.6 |
| Density | 5.67 |
| Melting Point | 733°C |
| Appearance | yellow-orange crystals |
13451-18-8 - Risk and Safety
| UN IDs | 1479 |
| Hazard Class | 5.1 |
| Packing Group | II |
13451-18-8 - References
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| production method | tellurium trioxide has two configurations: α-TeO 3 and β-TeO 3, the preparation method and properties of the two configurations of the three oxide are very different. The method of heating the original telluric acid dehydration can generate tellurium oxide: H 6 TeO 6 = TeO 3 3H 2 O place the pure Ortho tellurite H 6 TeO 6 into a porcelain crucible and slowly heat to 300~320 ℃, and stir continuously. When the water is completely removed and removed, it is cooled, and the product is leached several times with cold concentrated hydrochloric acid, from which the TeO 2 formed in the reaction can be removed. The product was then washed with water and finally dried at 100 °c. The product was obtained as α-TeO 3. The pure original telluric acid and a few drops of concentrated sulfuric acid were sealed in a glass tube, and heated at about 320 degrees C for 12~15H. The reaction product was then boiled in a concentrated KOH solution to remove α-TeO 3 therefrom. The residual β-TeO 3 was filtered off with a glass sand core funnel and washed thoroughly with water. Finally, after drying at 100 °c, the product was pure β-TeO 3. |
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: TELLURIUM TRIOXIDE Request for quotation
CAS: 13451-18-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 13451-18-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: TELLURIUM TRIOXIDE Request for quotation
CAS: 13451-18-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 13451-18-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
13451-18-8
Ethanol-18O
NITRICACID,8.0NSOLUTION
Methyl L-Pyroglutamate
4-溴-4'-羟基偶氮苯
氯吡格雷杂质29
(2E)-2-CYANO-3-[2,5-DIMETHYL-1-(TETRAHYDROFURAN-2-YLMETHYL)-1H-PYRROL-3-YL]ACRYLIC ACID
1142214-33-2
p-[2-Hydroxy-3-(isopropylamino)propoxy]benzaldehyde
Ethanol-18O
NITRICACID,8.0NSOLUTION
Methyl L-Pyroglutamate
4-溴-4'-羟基偶氮苯
氯吡格雷杂质29
(2E)-2-CYANO-3-[2,5-DIMETHYL-1-(TETRAHYDROFURAN-2-YLMETHYL)-1H-PYRROL-3-YL]ACRYLIC ACID
1142214-33-2
p-[2-Hydroxy-3-(isopropylamino)propoxy]benzaldehyde
