1251-85-0
4,4'-Diantipyrylmethane monohydrate
CAS: 1251-85-0
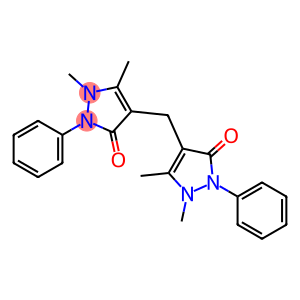
Molecular Formula: C23H24N4O2
1251-85-0 - Names and Identifiers
| Name | 4,4'-Diantipyrylmethane monohydrate |
| Synonyms | TOSLAB 33092 Diantipyrylmethane DIANTIPYRYLMETHANE DIANTIPYRRYLMETHANE Bisantipyrylmethane 4,4'-DIANTIPYRYLMETHANE Trichachnine monohydrate 4,4-Methylenediantipyrine 4,4'-METHYLENEDIANTIPYRINE 4,4'-Diantipyrylmethane monohydrate 4,4'-methylenebis[1,2-dihydro-1,5-dimethyl-2-phenyl-3h-pyrazol-3-on 4,4'-methylenebis(1,2-dihydro-1,5-dimethyl-2-phenyl)-3H-Pyrazol-3-one 4,4'-methanediylbis(1,5-dimethyl-2-phenyl-1,2-dihydro-3H-pyrazol-3-one) 4,4'-methanediylbis(1,5-dimethyl-2-phenyl-1,2-dihydro-3H-pyrazol-3-one) hydrate |
| CAS | 1251-85-0 |
| EINECS | 215-009-7 |
| InChI | InChI=1/C23H24N4O2.H2O/c1-16-20(22(28)26(24(16)3)18-11-7-5-8-12-18)15-21-17(2)25(4)27(23(21)29)19-13-9-6-10-14-19;/h5-14H,15H2,1-4H3;1H2 |
1251-85-0 - Physico-chemical Properties
| Molecular Formula | C23H24N4O2 |
| Molar Mass | 388.46 |
| Density | 1.1960 (rough estimate) |
| Melting Point | 156°C (dec.)(lit.) |
| Boling Point | 514.17°C (rough estimate) |
| Flash Point | 298.2°C |
| Water Solubility | 439mg/L(20 ºC) |
| Solubility | Insoluble in water, ether and alkali, soluble in acid, ethanol and chloroform. |
| Vapor Presure | 8.32E-14mmHg at 25°C |
| Appearance | White crystal |
| Color | White |
| BRN | 59702 |
| pKa | 1.26±0.65(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.6500 (estimate) |
| MDL | MFCD00149122 |
| Use | Used as a sensitive reagent for the determination of titanium and iron |
1251-85-0 - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29331990 |
1251-85-0 - Introduction
Used for the photometric determination of iridium, platinum, rhenium, titanium, bismuth, zinc, cobalt, iron, osmium, rhodium, thallium and tungsten. Gravimetric analysis of silicon.
Last Update:2022-10-16 17:12:58
1251-85-0 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| uses | as a sensitive reagent for the determination of titanium and iron as a sensitive developer for the determination of Au3, Ti4, Ir, iron (III), molybdenum, neodymium, uranium (VI), iridium, platinum, rhenium, etc. by spectrophotometry and extraction spectrophotometry. Weighing analysis to determine the precipitation agent of silicon. It can also be used as an extractant for various ions. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 4,4'-Methylenebis(1,5-dimethyl-2-phenyl-1H-pyrazol-3(2H)-one)... Visit Supplier Webpage Request for quotationCAS: 1251-85-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: DIANTIPYRYLMETHANE Request for quotation
CAS: 1251-85-0
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 1251-85-0
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Product Name: DIANTIPYRYLMETHANE Request for quotation
CAS: 1251-85-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1251-85-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: DAM Visit Supplier Webpage Request for quotationCAS: 1251-85-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4,4'-Methylenebis(1,5-dimethyl-2-phenyl-1H-pyrazol-3(2H)-one)... Visit Supplier Webpage Request for quotationCAS: 1251-85-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: DIANTIPYRYLMETHANE Request for quotation
CAS: 1251-85-0
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 1251-85-0
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Product Name: DIANTIPYRYLMETHANE Request for quotation
CAS: 1251-85-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1251-85-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: DAM Visit Supplier Webpage Request for quotationCAS: 1251-85-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



