1204580-89-1
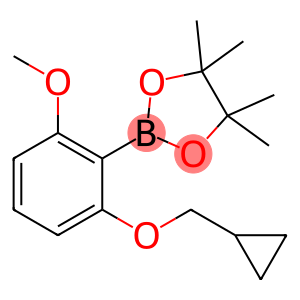
2-Cyclopropylmethoxy-6-methoxyphenylboronicacid pinacol ester
CAS: 1204580-89-1
Molecular Formula: C17H25BO4
1204580-89-1 - Names and Identifiers
1204580-89-1 - Physico-chemical Properties
| Molecular Formula | C17H25BO4 |
| Molar Mass | 304.19 |
| Storage Condition | Room Temprature |
| Sensitive | Irritant |
| MDL | MFCD13152001 |
1204580-89-1 - Introduction
pinacol, also known as pinacol, is an organic compound.
It is a white solid and soluble in some organic solvents such as ethanol and dimethyl sulfoxide. The compound contains phenyl and boron groups, which makes it have high reactivity and functionality.
Its use is mainly as an intermediate in organic synthesis. As a substrate, it can react with different functional groups for exchange reactions for the construction of more complex organic molecules. It is also often used as a ligand and used in organic synthesis reactions together with metal catalysts. In this way, specific chemical transformations can be achieved, such as carbon-carbon bond formation, carbon-oxygen bond formation, etc.
There are many methods for preparing pinacol ester. One common method is to react boric acid with the corresponding alcohol or ether.
Regarding safety information, the specific toxicity of this compound has not been clearly reported, but in general, organic compounds may cause irritation and sensitive reactions to humans. Therefore, during use, appropriate laboratory safety measures should be taken, such as wearing gloves, wearing protective glasses, and operating in a well-ventilated laboratory. At the same time, should avoid inhalation, intake and skin contact. If there is any discomfort, should be timely medical treatment.
It is a white solid and soluble in some organic solvents such as ethanol and dimethyl sulfoxide. The compound contains phenyl and boron groups, which makes it have high reactivity and functionality.
Its use is mainly as an intermediate in organic synthesis. As a substrate, it can react with different functional groups for exchange reactions for the construction of more complex organic molecules. It is also often used as a ligand and used in organic synthesis reactions together with metal catalysts. In this way, specific chemical transformations can be achieved, such as carbon-carbon bond formation, carbon-oxygen bond formation, etc.
There are many methods for preparing pinacol ester. One common method is to react boric acid with the corresponding alcohol or ether.
Regarding safety information, the specific toxicity of this compound has not been clearly reported, but in general, organic compounds may cause irritation and sensitive reactions to humans. Therefore, during use, appropriate laboratory safety measures should be taken, such as wearing gloves, wearing protective glasses, and operating in a well-ventilated laboratory. At the same time, should avoid inhalation, intake and skin contact. If there is any discomfort, should be timely medical treatment.
Last Update:2024-04-09 21:11:58
Supplier List
Product Name: 2-CyclopropylMethoxy-6-Methoxyphenylboronic acid pinacol ester Request for quotation
CAS: 1204580-89-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1204580-89-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2-CyclopropylMethoxy-6-Methoxyphenylboronic acid pinacol ester Request for quotation
CAS: 1204580-89-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1204580-89-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History
1204580-89-1
2-BROMO-1-CHLOROPROPANE SOLUTION 20,000UG/ML IN METHANOL 5ML
1-(4-aminophenyl)-N-{1-azabicyclo[2.2.2]octan-3-yl}methanesulfonamide
5-bromo-3,3-dimethyl-oxolane-2,4-dione
3-(溴甲基)-2-氟苯甲酸甲酯
(1-羟基-3-(甲基戊基氨基)丙叉)二膦酸单钠盐
2,3-Dibromobutanedioic acid dimethyl ester
70076-04-9
2H-Pyran-2-ol, tetrahydro-3,5-dimethyl-6-(1-methylethyl)-, [2R-(2α,3β,5α,6α)]- (9CI)
4-Hydroxy-5-(3-dimethylaminopropenoyl)benzenesulfonic acid chloride
2-BROMO-1-CHLOROPROPANE SOLUTION 20,000UG/ML IN METHANOL 5ML
1-(4-aminophenyl)-N-{1-azabicyclo[2.2.2]octan-3-yl}methanesulfonamide
5-bromo-3,3-dimethyl-oxolane-2,4-dione
3-(溴甲基)-2-氟苯甲酸甲酯
(1-羟基-3-(甲基戊基氨基)丙叉)二膦酸单钠盐
2,3-Dibromobutanedioic acid dimethyl ester
70076-04-9
2H-Pyran-2-ol, tetrahydro-3,5-dimethyl-6-(1-methylethyl)-, [2R-(2α,3β,5α,6α)]- (9CI)
4-Hydroxy-5-(3-dimethylaminopropenoyl)benzenesulfonic acid chloride
