115256-11-6
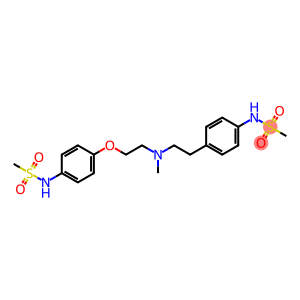
Dofetilide
CAS: 115256-11-6
Molecular Formula: C19H27N3O5S2
115256-11-6 - Names and Identifiers
115256-11-6 - Physico-chemical Properties
| Molecular Formula | C19H27N3O5S2 |
| Molar Mass | 441.56 |
| Density | 1.344±0.06 g/cm3(Predicted) |
| Melting Point | 147-1490C |
| Boling Point | 614.1±65.0 °C(Predicted) |
| Flash Point | 325.2°C |
| Solubility | DMSO: >20mg/mL |
| Vapor Presure | 5.13E-15mmHg at 25°C |
| Appearance | powder |
| Color | white to off-white |
| pKa | 7.0, 9.0, 9.6(at 25℃) |
| Storage Condition | 2-8°C |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
| Refractive Index | 1.613 |
| Physical and Chemical Properties | Crystallization from ethyl acetate-methanol (10:1), melting point 147~149 °c; Crystallization from hexane-ethyl acetate, melting point 151~152 °c; White crystalline solid has also been reported, melting point 161 °c. pKa 7.0,9.0,9.6. Partition coefficient (Ph = 7.4):0.96. |
| In vitro study | In Vitro culture of Xenopus oocytes, Dofetilide blocked HERG currents. In guinea pig myocardium, Dofetilide(1 μm) reduced the amplitude of IKR to 61% of the control current, as measured by a 200 MS test pulse and analysis of the deactivation tail current IKR. In intact canine hearts, Dofetilide increased apico-basal parallax, an increase in ERPs significantly more at the tip than at the basal. |
| In vivo study | In old myocardial infarction (MI) dogs, Dofetilide(100 mg/kg, I. P.) did not inhibit arrhythmias induced by two-stage coronary ligation and epinephrine or coronary ligation and reperfusion, but inhibition of reentry arrhythmias caused by PES. In dogs consuming digitalis arrhythmia, Dofetilide also showed antiarrhythmic effects. Dofetilide increases the QT interval and shows such a negative effect of other class III drugs, but differs from other classes in antiarrhythmic profiles with three agents, such as D-sotalol, E-4031, and in its MS-551 does not prevent the occurrence of ventricular fibrillation (VF) immediately after coronary reperfusion, and has certain antiarrhythmic effects on digitalis arrhythmia. In SD rats, Dofetilide causes an increase in resorption, and stage-dependent malformations. |
115256-11-6 - Risk and Safety
| Risk Codes | R61 - May cause harm to the unborn child R48/22 - Harmful danger of serious damage to health by prolonged exposure if swallowed. R51 - Toxic to aquatic organisms |
| Safety Description | S53 - Avoid exposure - obtain special instructions before use. S22 - Do not breathe dust. S36 - Wear suitable protective clothing. S57 - Use appropriate container to avoid environmental contamination. |
| WGK Germany | nwg |
| HS Code | 2935904000 |
115256-11-6 - Nature
Open Data Verified Data
from ethyl acetate methanol (10:1) crystallization, melting point 147~149 deg C; From hexane ethyl acetate crystallization, melting point 151~152 deg C; Also reported as white crystalline solid, melting point 161 °c. pK. 7.0,9.0,9.6. Partition coefficient (pH-7. 4):0.96.
115256-11-6 - Preparation Method
Open Data Verified Data
N-methyl -4-nitrophenylethylamine was dissolved in acetonitrile with 2-[4-nitrophenoxy] ethoxy bromide, potassium carbonate and sodium iodide were added, and the reaction was stirred under reflux. It is concentrated to dryness and the remaining oily solid is worked up to give the compound. The obtained compound was dissolved in ethanol and the reaction was catalyzed by Raney nickel. After filtration, the filtrate was concentrated to dryness. The resulting compound and methanesulfonic anhydride were dissolved in dichloromethane and stirred at room temperature. The product was further processed to obtain dofetilide.
115256-11-6 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.265 ml | 11.323 ml | 22.647 ml |
| 5 mM | 0.453 ml | 2.265 ml | 4.529 ml |
| 10 mM | 0.226 ml | 1.132 ml | 2.265 ml |
| 5 mM | 0.045 ml | 0.226 ml | 0.453 ml |
115256-11-6 - Use
Open Data Verified Data
developed by Pfizer Pharmaceuticals Inc. in the United States and marketed in 2000. Dofetilide is a relatively specific Class III antiarrhythmic drug, a selective potassium channel agonist. Treatment and prevention of atrial arrhythmias, such as atrial fibrillation, atrial flutter and paroxysmal supraventricular tachycardia, can also prevent the occurrence of ventricular tachycardia.
Supplier List
CAS: 115256-11-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 115256-11-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 115256-11-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 115256-11-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 115256-11-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 115256-11-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


