106861-44-3
Mivacurium chloride
CAS: 106861-44-3
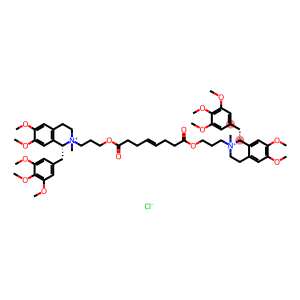
Molecular Formula: C58H80Cl2N2O14
106861-44-3 - Names and Identifiers
| Name | Mivacurium chloride |
| Synonyms | rac-Mivacron rac-BW-B 1090 rac-BW-B 1090U Mivacurium chloride MIVACURIUM CHLORIDE BW B1090U Dichloride rac Mivacurium Chloride 2,2'-{[(4E)-1,8-dioxooct-4-ene-1,8-diyl]bis(oxypropane-3,1-diyl)}bis[6,7-dimethoxy-2-methyl-1-(3,4,5-trimethoxybenzyl)-1,2,3,4-tetrahydroisoquinolinium] dichloride (1R,1'R)-2,2'-{[(4E)-1,8-Dioxooct-4-en-1,8-diyl]bis(oxypropan-3,1-diyl)}bis[6,7-dimethoxy-2-methyl-1-(3,4,5-trimethoxybenzyl)-1,2,3,4-tetrahydroisochinolinium]dichlorid [R-[R*,R*-(E)]]2,2'-(1,8-Dioxo-4-octene-1,8-diyl)bis(oxy-3,1-propanediyl)bis[1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-(3,4,5-trimethoxyphenyl)isoquinolinium] dichloride (1R,1'R)-2,2'-{[(4E)-1,8-Dioxooct-4-ene-1,8-diyl]bis(oxypropane-3,1-diyl)}bis[6,7-dimethoxy-2-methyl-1-(3,4,5-trimethoxybenzyl)-1,2,3,4-tetrahydroisoquinolinium] dichloride dichlorure de (1R,1'R)-2,2'-{[(4E)-1,8-dioxooct-4-ène-1,8-diyl]bis(oxypropane-3,1-diyl)}bis[6,7-diméthoxy-2-méthyl-1-(3,4,5-triméthoxybenzyl)-1,2,3,4-tétrahydroisoquinoléinium] [R-[R*,R*-(E)]]-2,2'-(1,8-Dioxo-4-octene-1,8-diyl)bis(oxy-3,1-propanediyl)bis(1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-[(3,4,5-trimethoxyphenyl)methyl]isoquinolinium) Dichloride rel-(1R,1'R)-2,2'-[[(4E)-1,8-Dioxo-4-octene-1,8-diyl]bis(oxy-3,1-propanediyl)]bis[1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-[(3,4,5-trimethoxyphenyl)methyl]- isoquinolinium Chloride isoquinolinium, 2,2'-[[(4E)-1,8-dioxo-4-octene-1,8-diyl]bis(oxy-3,1-propanediyl)]bis[1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-[(3,4,5-trimethoxyphenyl)methyl]-, dichloride, (1R,1'R)- |
| CAS | 106861-44-3 |
| EINECS | 643-006-4 |
| InChI | InChI=1/C58H80N2O14.2ClH/c1-59(25-21-41-35-47(63-3)49(65-5)37-43(41)45(59)29-39-31-51(67-7)57(71-11)52(32-39)68-8)23-17-27-73-55(61)19-15-13-14-16-20-56(62)74-28-18-24-60(2)26-22-42-36-48(64-4)50(66-6)38-44(42)46(60)30-40-33-53(69-9)58(72-12)54(34-40)70-10;;/h13-14,31-38,45-46H,15-30H2,1-12H3;2*1H/q+2;;/p-2/b14-13+; |
106861-44-3 - Physico-chemical Properties
| Molecular Formula | C58H80Cl2N2O14 |
| Molar Mass | 1100.17 |
| Specific Rotation(α) | 20D -62.7° (c = 1.9 in water) |
| Storage Condition | Hygroscopic, -20°C Freezer, Under Inert Atmosphere |
| Physical and Chemical Properties | Amorphous solid. [Α] D20-62.7 °(C = 1.9, water). |
| In vitro study | Mivacurium induces LAD2 cell degranulation in a dose-dependent manner. Mivacurium stimulates intracellular Ca 2+ influx in MRGPRX2-HEK293 cells but not in NC-HEK293 cells. Mivacurium induces the release of only low levels of mediators in LAD2 cells transfected with MRGPRX2-targeted small interfering siRNA. |
| In vivo study | Mivacurium causes pseudo-allergic reactions in C57 wild-type mice by inducing mast cells to release histamine and a decrease in body temperature. Mivacurium is rapidly hydrolyzed in the plasma and has a short duration of action (< 10 min). Mivacurium has many advantages, such as a rapid effect, nonneurological toxicity and a lack of heart rate alteration. |
106861-44-3 - Reference Information
| Biological activity | Mivacurium dichloride is a benzyl isoquinoline derivative, and is also a short-acting non-depolarizing neuromuscular blocker and skeletal muscle relaxant. Mivacurium dichloride can be coupled to nAChR to reduce or inhibit the depolarizing effect of acetylcholine on the terminal discs of muscle cells. |
| use | short-acting non-polarized neuromuscular blocker, short-acting muscle relaxant. It can be used for short-term surgical procedures, and can be used as an auxiliary drug for relaxing skeletal muscle, promoting tracheal intubation and general anesthesia during mechanical ventilation. |
| Production method | 5 ', 8-dimethoxy-N-methylpapaverine and 3-chloropropanol, in the presence of sodium iodide, reflux in 2-butanone. The obtained quaternary ammonium salt and octyl -4-alkylene diacyl chloride were reacted in dichloroethane at room temperature to obtain mikuronium chloride. |
Last Update:2024-04-09 21:21:28
Supplier List
Spot supply
Product Name: Mivacurium dichloride Visit Supplier Webpage Request for quotationCAS: 106861-44-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Mivacurium Chloride Visit Supplier Webpage Request for quotationCAS: 106861-44-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Mivacurium chloride Request for quotation
CAS: 106861-44-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 106861-44-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Mivacurium chloride Visit Supplier Webpage Request for quotation
CAS: 106861-44-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 106861-44-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Mivacurium dichloride Visit Supplier Webpage Request for quotationCAS: 106861-44-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Mivacurium Chloride Visit Supplier Webpage Request for quotationCAS: 106861-44-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Mivacurium chloride Request for quotation
CAS: 106861-44-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 106861-44-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Mivacurium chloride Visit Supplier Webpage Request for quotation
CAS: 106861-44-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 106861-44-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



