10257-54-2
Copper sulfate monohydrate
CAS: 10257-54-2
Molecular Formula: CuH2O5S
10257-54-2 - Names and Identifiers
10257-54-2 - Physico-chemical Properties
| Molecular Formula | CuH2O5S |
| Molar Mass | 177.62 |
10257-54-2 - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R36/38 - Irritating to eyes and skin. R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S22 - Do not breathe dust. S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3077 |
10257-54-2 - Reference Information
| food additive maximum allowable usage maximum allowable residue standard | ▼ ▲ additive Chinese name food Chinese name additive function maximum allowable usage amount (g/kg) maximum allowable residue amount (g/kg) copper: copper sulfate [minerals] drinking liquid nutrition fortifier 4~5mg(1. strengthening amount based on elemental copper: dairy products, infant formula food 3~4 mg/kg drinking liquid 1~1.25 mg/kg 2. copper element content in various copper salts: copper carbonate 54%, copper sulfate (5 crystal water) 25.5%, anhydrous copper sulfate 39.8%) copper: copper sulfate [minerals] infant food nutrition fortifier 7.5~10mg(1. fortified amount based on elemental copper: dairy products, infant formula food 3~4 mg/kg, drinking liquid 1~1.25 mg/kg 2. copper content in various copper salts: copper carbonate 54%, copper sulfate (5 crystal water) 25.5%, anhydrous copper sulfate 39.8%) copper: copper sulfate [minerals] dairy nutrition fortifier 12~16mg(1. fortification amount based on elemental copper: dairy products, infant formula food for 3~4 mg/kg, drinking liquid for 1~1.25 mg/kg 2. copper content in various copper salts: copper carbonate 54%, copper sulfate (5 crystal water) 25.5%, anhydrous copper sulfate 39.8%) |
| production method | 1. copper powder is roasted at 600~700 ℃ by sulfuric acid method, oxidized into copper oxide, then decomposed by sulfuric acid, clarified to remove insoluble impurities, cooled, crystallized, filtered and dried, the finished copper sulfate product is produced. The reaction formula is as follows. 2. The waste electrolyte of electrolyte recovery method (containing Cu50 ~ 60g/L,H2SO4180 ~ 200g/L) reacts with the roasted copper mud to make fine copper powder. After the reaction liquid is separated and settled, the clear liquid is cooled, crystallized, separated and dried to obtain the finished copper sulfate product. 3. The copper compound precipitate in the copper ammonium acetate solution in the copper washing tower when the synthetic ammonia feed gas of the nitrogen fertilizer plant is recovered by the recovery method. After roasting at 700 ℃, it is oxidized to copper oxide and then reacts with sulfuric acid to generate copper sulfate. 4. The chemical concentration crystallization method uses low-grade copper oxide ore, pulverizes to a certain particle size, adds sulfuric acid to impregnate, adds copper-soluble iron precipitation agent, obtains a copper sulfate solution with a copper-iron ratio greater than 100, and then adds a chemical concentrator for concentration, and drains 70% ~ 90% of water. Finally, it is evaporated, cooled, crystallized, separated, and air-dried to obtain a finished copper sulfate product. |
| EPA chemical information | information provided by: ofmpub.epa.gov (external link) |
Last Update:2024-04-09 02:00:00
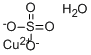
Supplier List
Product Name: Cupric sulfate monohydrate Request for quotation
CAS: 10257-54-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 10257-54-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Cupric sulfate monohydrate Request for quotation
CAS: 10257-54-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 10257-54-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History
