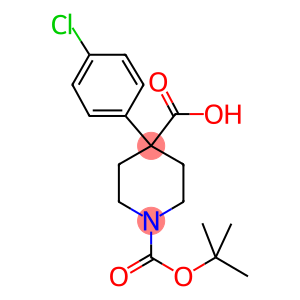
1-Boc-4-(4-chlorophenyl)-4-carboxypiperidine
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester
CAS: 644981-94-2
Molecular Formula: C17H22ClNO4
1-Boc-4-(4-chlorophenyl)-4-carboxypiperidine - Names and Identifiers
1-Boc-4-(4-chlorophenyl)-4-carboxypiperidine - Physico-chemical Properties
| Molecular Formula | C17H22ClNO4 |
| Molar Mass | 339.81 |
| Storage Condition | Room Temprature |
1-Boc-4-(4-chlorophenyl)-4-carboxypiperidine - Risk and Safety
| HS Code | 29333990 |
1-Boc-4-(4-chlorophenyl)-4-carboxypiperidine - Introduction
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester(1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester is also known as a BOC-DPC BOC-DPC-Cl.
Nature:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester is a white or off-white crystalline powder. Its molecular formula is C18H23ClNO4 and its relative molecular mass is 353.83g/mol. It is a stable solid, not easily soluble in water, but soluble in organic solvents such as dimethylformamide and chloroform.
Use:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester are mainly used as intermediates in organic synthesis. It can be used to synthesize new drug molecules, drug precursors, pesticides and heterocyclic compounds. The BOC protecting group of the compound can protect other functional groups during the synthesis.
Preparation Method:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester can be prepared by reacting 4-piperidinemethosulfate with a 1,4-dichlorobutane derivative. The preparation involves the synthesis of the piperidine ring and the substituted phenyl group, followed by esterification of the acid.
Safety Information:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophyll)-,1-(1,1-dimenthylethyl) ester is less toxic, but it is still necessary to maintain appropriate laboratory safety operations. Wear appropriate protective equipment during operation to avoid inhalation, skin contact and eating. In case of accidental contact or inhalation, the affected area should be cleaned promptly and medical advice should be sought if necessary.
Nature:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester is a white or off-white crystalline powder. Its molecular formula is C18H23ClNO4 and its relative molecular mass is 353.83g/mol. It is a stable solid, not easily soluble in water, but soluble in organic solvents such as dimethylformamide and chloroform.
Use:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester are mainly used as intermediates in organic synthesis. It can be used to synthesize new drug molecules, drug precursors, pesticides and heterocyclic compounds. The BOC protecting group of the compound can protect other functional groups during the synthesis.
Preparation Method:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophenyl)-,1-(1,1-dimethylethyl) ester can be prepared by reacting 4-piperidinemethosulfate with a 1,4-dichlorobutane derivative. The preparation involves the synthesis of the piperidine ring and the substituted phenyl group, followed by esterification of the acid.
Safety Information:
1,4-Piperidinedicarboxylic acid, 4-(4-chlorophyll)-,1-(1,1-dimenthylethyl) ester is less toxic, but it is still necessary to maintain appropriate laboratory safety operations. Wear appropriate protective equipment during operation to avoid inhalation, skin contact and eating. In case of accidental contact or inhalation, the affected area should be cleaned promptly and medical advice should be sought if necessary.
Last Update:2024-04-09 02:00:47
Supplier List
Product Name: N-BOC-4-(P-CHLOROPHENYL)-4-PIPERIDINE CARBOXYLIC ACID Request for quotation
CAS: 644981-94-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 644981-94-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: N-BOC-4-(P-CHLOROPHENYL)-4-PIPERIDINE CARBOXYLIC ACID Visit Supplier Webpage Request for quotation
CAS: 644981-94-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 644981-94-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: N-BOC-4-(P-CHLOROPHENYL)-4-PIPERIDINE CARBOXYLIC ACID Request for quotation
CAS: 644981-94-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 644981-94-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: N-BOC-4-(P-CHLOROPHENYL)-4-PIPERIDINE CARBOXYLIC ACID Visit Supplier Webpage Request for quotation
CAS: 644981-94-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 644981-94-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

