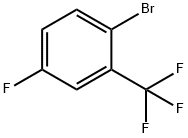
1-BROMO-2-(TRIFLUOROMETHYL)-4-FLUOROBENZENE
2-bromo-5-fluorobenzotrifluoride
CAS: 40161-55-5
Molecular Formula: C7H3BrF4
1-BROMO-2-(TRIFLUOROMETHYL)-4-FLUOROBENZENE - Names and Identifiers
1-BROMO-2-(TRIFLUOROMETHYL)-4-FLUOROBENZENE - Physico-chemical Properties
| Molecular Formula | C7H3BrF4 |
| Molar Mass | 243 |
| Density | 1.695g/mLat 25°C(lit.) |
| Boling Point | 136-143°C(lit.) |
| Flash Point | 147°F |
| Vapor Presure | 3.1mmHg at 25°C |
| BRN | 2643544 |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.465(lit.) |
1-BROMO-2-(TRIFLUOROMETHYL)-4-FLUOROBENZENE - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S37 - Wear suitable gloves. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| HS Code | 29039990 |
| Hazard Class | IRRITANT |
1-BROMO-2-(TRIFLUOROMETHYL)-4-FLUOROBENZENE - Reference Information
| introduction | 2-bromo-5-fluorotrifluoro-toluene is a colorless liquid under normal temperature and pressure. it is a commercial organic synthon with good solubility in common organic solvents such as methylene chloride, ethyl acetate and dimethyl sulfoxide, but its solubility in water is poor. It can be used as an intermediate in organic synthesis. |
| use | 2-bromo -5-fluorotrifluorotoluene is a light yellow liquid and can be used as an important medicine, pesticide and new liquid crystal material intermediate. For example, it is an important intermediate for the synthesis of anti-tumor drug bicalutamide, bicalutamide is an anti-androgen non-steroidal steroid, it is well tolerated and has no obvious side effects after taking it. With the continuous expansion of the use of 2-bromo-5-fluorotrifluorotoluene, the market demand is also increasing. |
| synthesis method | for the synthesis of 2-bromo -5-fluorotrifluorotoluene, the conventional synthesis method is starting from 3-fluorotrifluorotoluene, and the commonly used brominating reagent is liquid bromine by brominating position 5 of benzene ring under appropriate conditions. According to literature research, the reaction has good regioselectivity and high reaction efficiency. |
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
Last Update:2024-04-09 19:05:03
Supplier List
Spot supply
Product Name: 2-Bromo-5-fluorobenzotrifluoride Visit Supplier Webpage Request for quotationCAS: 40161-55-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-bromo-5-fluorobenzotrifluoride Request for quotation
CAS: 40161-55-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 40161-55-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-Bromo-5-fluorobenzotrifluoride Visit Supplier Webpage Request for quotationCAS: 40161-55-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-Bromo-5-fluorobenzotrifluoride Request for quotation
CAS: 40161-55-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 40161-55-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Bromo-5-fluorobenzotrifluoride Visit Supplier Webpage Request for quotationCAS: 40161-55-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-bromo-5-fluorobenzotrifluoride Request for quotation
CAS: 40161-55-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 40161-55-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-Bromo-5-fluorobenzotrifluoride Visit Supplier Webpage Request for quotationCAS: 40161-55-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-Bromo-5-fluorobenzotrifluoride Request for quotation
CAS: 40161-55-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 40161-55-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
View History



