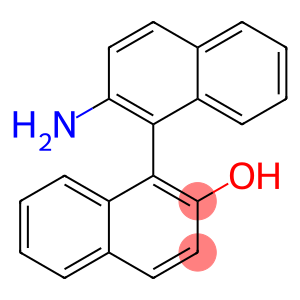
1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol
(S)-2'-Amino-1,1'-binaphthalen-2-ol
CAS: 137848-29-4
Molecular Formula: C20H15NO
1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol - Names and Identifiers
| Name | (S)-2'-Amino-1,1'-binaphthalen-2-ol |
| Synonyms | S-Nobin (S)-(-)-Nobin S-2'-Amino-1,1'-Binaphthalen-2-Ol (S)-2'-Amino-1,1'-binaphthalen-2-ol (S)-2-Amino-2'-Hydroxy-1,1'-Binaphthyl (S)-(-)-2-AMINO-2'-HYDROXY-1 1'-BINAPHT S)-(-)-2-Amino-2'-Hydroxy-1,1'-Binaphthol 1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol (S)-2-Amino-2'-Hydroxy-1,1'-Dinaphthalene S-2'-Amino-1,1'-binaphthalen-2-ol, S-NOBIN (S)-(-)-2-Amino-2'-hydroxy-1,1'-binaphthalene (S)-(-)-1-(2-Amino-1-naphthyl)-2-naphthol(S)-(-)-NOBIN(S)-(-)-2'-Amino-1,1'-binaphthalen-2-ol |
| CAS | 137848-29-4 |
1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol - Physico-chemical Properties
| Molecular Formula | C20H15NO |
| Molar Mass | 285.34 |
| Density | 1.275 |
| Melting Point | 171.0 to 175.0 °C |
| Boling Point | 471.5±30.0 °C(Predicted) |
| Appearance | White to light red powder or crystal |
| Color | White to Light red to Green |
| pKa | 8.90±0.50(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Sensitive | Argon filling operation and storage |
| MDL | MFCD01882346 |
1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S60 - This material and its container must be disposed of as hazardous waste. S39 - Wear eye / face protection. |
| UN IDs | UN 3077 9 / PGIII |
| WGK Germany | 3 |
| HS Code | 29222990 |
1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol - Reference Information
| Application prospects | (S)-( )-2-amino -2 '-hydroxy -1,1'-binaphthalene is a common chiral synthesis reagent. NOBIN(2-amino -2 '-hydroxy -1,1'-binaphthalene) is an organic molecule used for asymmetric catalysis. NOBIN is associated with BINOL(1,1 ′-li-2, 2 ′-naphthol, abbreviated as 2-amino-2'-hydroxy -1,1 ′-binaphthalene) and other analogs, with both chiral axes and scaffolds for certain chemical reactions. NOBIN is an excellent catalyst for aldol reaction, which can produce reliable products, good yields and excellent diastereoselectivity. Although the rotation of the bond around the connecting ring is limited by the hydrogen atom, the enantiomeric pure NOBIN may be racemic when heated. Many chiral catalysts or metal catalyst ligands have been synthesized using 2-amino -2 '-hydroxy -1,1'-binaphthalene, and used for Diels-Alder reactions, Aldol condensation, alkylation of carbonyl compounds, reduction of latent chiral ketones, Michael addition and asymmetric epoxidation, showing good catalytic activity and enantioselectivity, showing broad application prospects. |
| preparation | since the 70 s, more and more attention has been paid to the synthesis and resolution of racemic 2-amino -2 '-hydroxy -1,1'-binaphthalene and the asymmetric synthesis of 2-amino -2 '-hydroxy -1,1'-binaphthalene. The racemate 2-amino -2 '-hydroxy -1,1'-binaphthalene is basically obtained by the oxidative coupling of 2-naphthol and 2-naphthylamine, and the optically active body is often obtained by the resolution method. There are biological and chemical methods for the resolution of such compounds. The chemical resolution method uses the characteristics of the functional group of 2-amino -2 '-hydroxy -1,1'-binaphthalene to react with photoactive reagents to obtain diastereomers, and then use their properties to purify and decompose to obtain photoactive substances. In addition, enzyme and chromatographic methods are used for resolution. The resolution of 2-amino -2 '-hydroxy -1,1'-binaphthalene has been studied and reviewed. The synthesis of 2-amino -2 '-hydroxy -1,1'-binaphthalene is almost always obtained by oxidizing 2-naphthol and 2-naphthylamine with suitable oxidants. (S)-( )-2-amino -2 '-hydroxy -1,1'-binaphthalene synthesis reaction formula is as follows: fig. 1 (S)-( )-2-amino -2 '-hydroxy -1,1'-binaphthalene synthesis reaction formula |
| Uses | Various chiral synthetic reagents. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: (S)-(-)-2-Amino-2'-hydroxy-1,1'-binaphthyl Visit Supplier Webpage Request for quotationCAS: 137848-29-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: O-(2-Aminoethyl)-L-Serine Hydrochloride Request for quotation
CAS: 137848-29-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 137848-29-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (S)-2'-Amino-1,1'-binaphthalen-2-ol Request for quotation
CAS: 137848-29-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 137848-29-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: (S)-(-)-2-Amino-2'-hydroxy-1,1'-binaphthyl Visit Supplier Webpage Request for quotationCAS: 137848-29-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-(-)-2-Amino-2'-hydroxy-1,1'-binaphthyl Visit Supplier Webpage Request for quotationCAS: 137848-29-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: O-(2-Aminoethyl)-L-Serine Hydrochloride Request for quotation
CAS: 137848-29-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 137848-29-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (S)-2'-Amino-1,1'-binaphthalen-2-ol Request for quotation
CAS: 137848-29-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 137848-29-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: (S)-(-)-2-Amino-2'-hydroxy-1,1'-binaphthyl Visit Supplier Webpage Request for quotationCAS: 137848-29-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
1-(2-aMinonaphthalen-1-yl)naphthalen-2-ol
6-Bromo-4,4-dimethylthiochroman
3-氟-2-碘异烟酸
1260897-95-7
N'-[(1,2-dimethyl-1H-indol-3-yl)methylene]-2-{2-nitrophenoxy}acetohydrazide
4-氨基-1-哌啶甲酸乙酯,N-乙氧羰基-4-氨基哌啶
2,3,5,6,7,8,9,9b-Octahydro-5,5-dimethyl-1H-cyclopent[g]indolizine
Bicyclo[3.1.0]hexan-3-one, 4-hydroxy-4-methyl-1-(1-methylethyl)-, (1S,4R,5S)-
Methyl 3-keto-delta-4-cholate
Formaldehyde,polymer with 4-(1,1-dimethylethyl)phenol and 4-octylphenol
6-Bromo-4,4-dimethylthiochroman
3-氟-2-碘异烟酸
1260897-95-7
N'-[(1,2-dimethyl-1H-indol-3-yl)methylene]-2-{2-nitrophenoxy}acetohydrazide
4-氨基-1-哌啶甲酸乙酯,N-乙氧羰基-4-氨基哌啶
2,3,5,6,7,8,9,9b-Octahydro-5,5-dimethyl-1H-cyclopent[g]indolizine
Bicyclo[3.1.0]hexan-3-one, 4-hydroxy-4-methyl-1-(1-methylethyl)-, (1S,4R,5S)-
Methyl 3-keto-delta-4-cholate
Formaldehyde,polymer with 4-(1,1-dimethylethyl)phenol and 4-octylphenol



