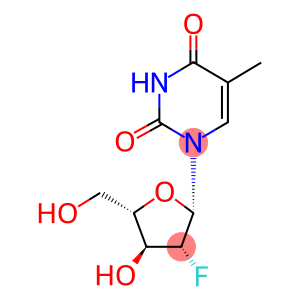
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione
CAS: 163252-36-6
Molecular Formula: C10H13FN2O5
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione - Names and Identifiers
| Name | 1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione |
| Synonyms | Levovir Clevudine CLEVUDINE 2'-Fluoro-5-methylarabinosyluracil 1-(2''-DEOXY-2''-FLUORO--L-ARABINOFURANOSYL)-5-METHYLURACIL 1-(2'-DEOXY-2'-FLUORO-BETA-L-ARABINOFURANOSYL)-5-METHYLURACIL 1-[(2S,3R,4S,5S)-3-Fluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione 1-[(2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-5-methyl-pyrimidine-2,4-dione 1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione |
| CAS | 163252-36-6 |
| EINECS | 200-001-2 |
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione - Physico-chemical Properties
| Molecular Formula | C10H13FN2O5 |
| Molar Mass | 260.22 |
| Density | 1.55±0.1 g/cm3(Predicted) |
| Melting Point | 184-185° |
| Specific Rotation(α) | D25 -111.77° (c = 0.23 in methanol) |
| Solubility | DMSO : 50 mg/mL mother liquor preservation: sub-package and freeze storage to avoid repeated freezing and thawing;-20 ℃,1 month;-80 ℃,6 months (after dilution, the solution temperature is low and storage may precipitate, try to use it now) Cell experiment: Dissolve with DMSO first: dilute with culture medium then, and the dilution process is recommended to be carried out in stages to avoid too fast concentration change leading to compound precipitation. If the compound is precipitated during the dilution process, it can be redissolved by ultrasound. During dilution, ensure that the final concentration of DMSO in the working fluid should be below 0.1% as far as possible, and the maximum should not exceed 0.5%, and set up a DMSO control group with corresponding concentration. Animal experiment: Dissolve with DMSO first: dilute with water or normal saline, etc. The dilution process is recommended to be carried out in sections to avoid excessive concentration changes leading to compound p |
| Appearance | Solid |
| Color | White |
| pKa | 9.55±0.10(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Use | Clevudine, also known as L-FMAU, is an antiviral used in the treatment of Hepatitis B. Clevudine is differentiated from other oral agents by an unusual activation pathway to the biochemically active triphosphate, a mechanism of action of clevudine triphosphate that inhibits multiple steps of the hepatitis B virus (HBV) intracellular life cycle, a long half-life, and significant reduction of covalently closed circular DNA (cccDNA) in animal models. |
| Target | DNA polymerase ;RNA polymerase |
| In vitro study | Clevudine is a potent anti-HBV agent (EC 50 0.1 μm in HepG22.2.15 cells) as well as an anti-EB virus agent, which has been shown to be effective in a variety of cell lines, including MT2,CEM, both H1 and HepG22.2.15 and bone marrow progenitor cells had low cytotoxicity. Clevudine is metabolized by the cell by thymidine kinase and deoxycytidine kinase in the cell to the phosphate form, and subsequently diphosphate and triphosphate. Clevudine is known to act specifically on viral DNA synthesis, and its triphosphate inhibits HBV DNA synthesis in a dose-dependent manner without being incorporated into DNA or chain termination. Clevudine causes an increase in diphosphate and triphosphate metabolites in these analogs. Clevudine monophosphate (L-FMAUMP) is the weaker ligand of the D-isomer. Clevudine is readily phosphorylated to the 5 '-triphosphate form compound in cell culture, which is involved in the mechanism of action of Clevudine. |
| In vivo study | Clevudine caused a delay in viral relapse in a dose-dependent manner, reducing the loss of covalently closed circular DNA in the Woodchuck Model. Clevudine(10 mg/kg, IP) in combination with Emtricitabine (30 mg/kg, IP) resulted in a rapid and significant inhibition of virus replication in woodchucks, serum WHV DNA decreased by 10 to the 4th power. In WHV-infected marmots, Clevudine(10 mg/kg, I. P.) versus Emtricitabine (30 mg/kg, IP.) the combination results in an effective and long-lasting anti-hepatotropic effect in the liver and blood. |
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione - Risk and Safety
| Toxicity | LD50 in mice, rats (mg/kg): >5000, >3000 orally (Painter) |
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione - Reference
| Reference Show more | 1: Anderson DL. Clevudine for hepatitis B. Drugs Today (Barc). 2009 May;45(5):331-50. doi: 10.1358/dot.2009.45.5.1354223. Review. PubMed PMID: 19584963. 2: Lau GK, Leung N. Forty-eight weeks treatment with clevudine 30 mg qd versus lamivudine 100 mg qd for chronic hepatitis B infection: a double-blind randomized study. Korean J Hepatol. 2010 Sep;16(3):315-20. doi: 10.3350/kjhep.2010.16.3.315. PubMed PMID: 20924215; PubMed Central PMCID: PMC3304600. 3: Kocic I. Clevudine University of Georgia/Abbott/Bukwang/Triangle/Yale University. Curr Opin Investig Drugs. 2000 Nov;1(3):308-13. Review. PubMed PMID: 11249713. 4: Yoo BC, Kim JH, Kim TH, Koh KC, Um SH, Kim YS, Lee KS, Han BH, Chon CY, Han JY, Ryu SH, Kim HC, Byun KS, Hwang SG, Kim BI, Cho M, Yoo K, Lee HJ, Hwang JS, Kim YS, Lee YS, Choi SK, Lee YJ, Yang JM, Park JW, Lee MS, Kim DG, Chung YH, Cho SH, Choi JY, Kweon YO, Lee HY, Jeong SH, Yoo HW, Lee HS. Clevudine is highly efficacious in hepatitis B e antigen-negative chronic hepatitis B with durable off-therapy viral suppression. Hepatology. 2007 Oct;46(4):1041-8. PubMed PMID: 17647293. 5: Asselah T, Lada O, Moucari R, Marcellin P. Clevudine: a promising therapy for the treatment of chronic hepatitis B. Expert Opin Investig Drugs. 2008 Dec;17(12):1963-74. doi: 10.1517/13543780802535760 . Review. PubMed PMID: 19012511. 6: Hui CK, Lau GK. Clevudine for the treatment of chronic hepatitis B virus infection. Expert Opin Investig Drugs. 2005 Oct;14(10):1277-84. Review. PubMed PMID: 16185170. |
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 3.843 ml | 19.215 ml | 38.429 ml |
| 5 mM | 0.769 ml | 3.843 ml | 7.686 ml |
| 10 mM | 0.384 ml | 1.921 ml | 3.843 ml |
| 5 mM | 0.077 ml | 0.384 ml | 0.769 ml |
Last Update:2024-01-02 23:10:35
1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione - Reference Information
| effect | the mechanism of action of Claudine is different from LAM and adefovir. Because of its 3 'position on the hydroxyl group, the formation of triphosphate L-FM AU-TP binding in the active site, induced polymerase conformation changes, so that the synthesis of Virus DNA chain can not proceed smoothly. |
| biological activity | Clevudine is an antiviral drug used to treat hepatitis B. |
| Target | Value |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: Clevudine Visit Supplier Webpage Request for quotationCAS: 163252-36-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione Request for quotation
CAS: 163252-36-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 163252-36-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Clevudine Request for quotation
CAS: 163252-36-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 163252-36-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: 1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione Visit Supplier Webpage Request for quotationCAS: 163252-36-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Clevudine Visit Supplier Webpage Request for quotationCAS: 163252-36-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione Request for quotation
CAS: 163252-36-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 163252-36-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Clevudine Request for quotation
CAS: 163252-36-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 163252-36-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: 1-((2S,3R,4S,5S)-3-fluoro-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione Visit Supplier Webpage Request for quotationCAS: 163252-36-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



