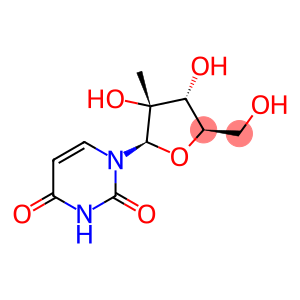
1-((2R,3R,4R,5R)-TETRAHYDRO-3,4-DIHYDROXY-5-(HYDROXYMETHYL)-3-METHYLFURAN-2-YL)PYRIMIDINE-2,4(1H,3H)-DIONE
2'-C-Methyluridine
CAS: 31448-54-1
Molecular Formula: C10H14N2O6
1-((2R,3R,4R,5R)-TETRAHYDRO-3,4-DIHYDROXY-5-(HYDROXYMETHYL)-3-METHYLFURAN-2-YL)PYRIMIDINE-2,4(1H,3H)-DIONE - Names and Identifiers
| Name | 2'-C-Methyluridine |
| Synonyms | EOS-61181 2'-C-Methyluridine Uridine, 2'-C-Methyl- 2'-C-Methyl-1-β-D-ribofuranosyluracil 1-(2-C-Methyl-β-D-ribofuranosyl)uracil 1-((2R,3R,4R,5R)-3,4-Dihydroxy-5-(hydroxyMethyl)-3-Methyltetrahydrofuran-2-yl)pyriMidine-2,4(1H,3H)-dione 1-((2R,3R,4R,5R)-TETRAHYDRO-3,4-DIHYDROXY-5-(HYDROXYMETHYL)-3-METHYLFURAN-2-YL)PYRIMIDINE-2,4(1H,3H)-DIONE 1-[(2R,3R,4R,5R)-3,4-dihydroxy-5-(hydroxymethyl)-3-methyloxolan-2-yl]-1,2,3,4-tetrahydropyrimidine-2,4-dione |
| CAS | 31448-54-1 |
1-((2R,3R,4R,5R)-TETRAHYDRO-3,4-DIHYDROXY-5-(HYDROXYMETHYL)-3-METHYLFURAN-2-YL)PYRIMIDINE-2,4(1H,3H)-DIONE - Physico-chemical Properties
| Molecular Formula | C10H14N2O6 |
| Molar Mass | 258.23 |
| Density | 1.572 |
| Melting Point | 110-112 °C |
| Appearance | powder to crystal |
| Color | White to Almost white |
| pKa | 9.39±0.10(Predicted) |
| Storage Condition | 2-8°C |
1-((2R,3R,4R,5R)-TETRAHYDRO-3,4-DIHYDROXY-5-(HYDROXYMETHYL)-3-METHYLFURAN-2-YL)PYRIMIDINE-2,4(1H,3H)-DIONE - Reference Information
| background | nucleosides and their derivatives are effective in the treatment of malignant tumors and antiviral effects, of which 2 '-C-methyluridine is the synthesis of methylcytidine The key intermediate of drugs. Using D-ribose as the starting material, 1-O-acetyl -2,3,5-tri-O-benzoyl-β-D-baranyose was obtained by a series reaction of methylamine, benzoyl and acetylation. The optimal reaction conditions were determined by single-factor experiments in each step, and the yield reached 70.6%. Subsequently, the intermediate Danhongli, oxygen 1 methylation, and benzoylation reactions obtained the glycosyl part of 2 '-C-methyluridine. |
| synthesis method | u6 and uricidine are coupled in the presence of silane reagents to form a uridine structure with a protective group, and then the protective group is removed to obtain the target product 22-C-methyluridine (u1), which is currently the most common route for the synthesis of nucleoside drugs. |
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 1-(2-C-Methyl-β-D-ribofuranosyl)uracil Visit Supplier Webpage Request for quotationCAS: 31448-54-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2'-C-Methyluridine Request for quotation
CAS: 31448-54-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31448-54-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2'-C-Methyluridine Request for quotation
CAS: 31448-54-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 31448-54-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2'-C-Methyluridine Visit Supplier Webpage Request for quotation
CAS: 31448-54-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 31448-54-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-(2-C-Methyl-β-D-ribofuranosyl)uracil Visit Supplier Webpage Request for quotationCAS: 31448-54-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2'-C-Methyluridine Request for quotation
CAS: 31448-54-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31448-54-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2'-C-Methyluridine Request for quotation
CAS: 31448-54-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 31448-54-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2'-C-Methyluridine Visit Supplier Webpage Request for quotation
CAS: 31448-54-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 31448-54-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
1-((2R,3R,4R,5R)-TETRAHYDRO-3,4-DIHYDROXY-5-(HYDROXYMETHYL)-3-METHYLFURAN-2-YL)PYRIMIDINE-2,4(1H,3H)-DIONE
Dabigatran Etexilate Impurity U
Bicyclo[4.2.1]nona-2,4-diene, 7-methoxy-, exo- (9CI)
2-Chloro-3-(phenylmethyl)pyrazine
PILARALISIB (XL147)试剂
3-(2,3-Dihydro-1h-inden-5-yl)-2,2-dimethylpropanoic acid
7-(tert-butoxycarbonyl)-7-aza-bicyclo[2.2.1]heptene-2-carboxylic acid methyl ester racemate
尼达尼布杂质
(2R,3R,4R,5R)-2-(4-氨基-7H-吡咯并[2,3-d]嘧啶-7-基)-5-(羟甲基)-3-甲基四氢呋喃-3,4-二醇
BUTANOIC ACID, 3-METHYL-,3-HEXENYL ESTER, (Z)-
Dabigatran Etexilate Impurity U
Bicyclo[4.2.1]nona-2,4-diene, 7-methoxy-, exo- (9CI)
2-Chloro-3-(phenylmethyl)pyrazine
PILARALISIB (XL147)试剂
3-(2,3-Dihydro-1h-inden-5-yl)-2,2-dimethylpropanoic acid
7-(tert-butoxycarbonyl)-7-aza-bicyclo[2.2.1]heptene-2-carboxylic acid methyl ester racemate
尼达尼布杂质
(2R,3R,4R,5R)-2-(4-氨基-7H-吡咯并[2,3-d]嘧啶-7-基)-5-(羟甲基)-3-甲基四氢呋喃-3,4-二醇
BUTANOIC ACID, 3-METHYL-,3-HEXENYL ESTER, (Z)-



