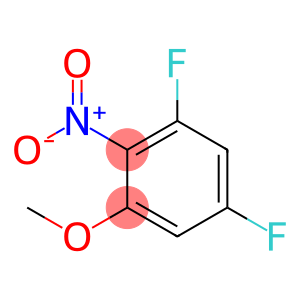
1,5-difluoro-3-METHOXY-2-NITRO-BENZENE
3,5-difluoro-2-nitroanisole
CAS: 66684-61-5
Molecular Formula: C7H5F2NO3
1,5-difluoro-3-METHOXY-2-NITRO-BENZENE - Names and Identifiers
1,5-difluoro-3-METHOXY-2-NITRO-BENZENE - Physico-chemical Properties
| Molecular Formula | C7H5F2NO3 |
| Molar Mass | 189.12 |
| Density | 1.414 |
| Boling Point | 238℃ |
| Flash Point | 98℃ |
| Storage Condition | Sealed in dry,Room Temperature |
1,5-difluoro-3-METHOXY-2-NITRO-BENZENE - Introduction
3,5-difluoro-2-nitroanisole(3,5-difluoro-2-nitroanisole) is an organic compound. Its chemical formula is C7H5F2NO3 and its molecular weight is 189.11g/mol.
Its properties are as follows:
-Appearance: Colorless crystalline solid
-Melting point: 62-64 degrees Celsius
-Boiling point: 290-293 degrees Celsius
-Solubility: Slightly soluble in water, better solubility in organic solvents
It has special uses in some areas:
-Chemical synthesis: can be used as an important intermediate in organic synthesis
-Pharmaceutical field: It has certain biological activity and can be used as a precursor for drug development or synthetic drugs
-Pesticide field: can be used for the synthesis of certain pesticides
There are many methods for preparing 3,5-difluororo-2-nitroanisole. The following is a general synthetic route:
First, toluene is reacted with nitric acid to obtain 2-nitrotoluene. Then, 2-nitrotoluene is reacted with hydrogen fluoride under appropriate reaction conditions to obtain 1,5-difluoro-2-nitrotoluene. Finally, 1,5-difluoro-2-nitrotoluene is reacted with methyl ether in the presence of hydrogen iodide to give 3,5-difluoro-2-nitroanisole.
Regarding safety information, 3,5-difluoro-2-nitroanisole is a chemical and needs to be stored and handled correctly. During use, safety procedures must be followed, such as wearing appropriate personal protective equipment (such as gloves, goggles and laboratory coats), avoiding direct contact with the skin and inhaling its vapors. In addition, attention should also be paid to fire and explosion-proof measures to ensure safe use. In the event of an accident, emergency measures shall be taken immediately and professional advice shall be consulted.
Its properties are as follows:
-Appearance: Colorless crystalline solid
-Melting point: 62-64 degrees Celsius
-Boiling point: 290-293 degrees Celsius
-Solubility: Slightly soluble in water, better solubility in organic solvents
It has special uses in some areas:
-Chemical synthesis: can be used as an important intermediate in organic synthesis
-Pharmaceutical field: It has certain biological activity and can be used as a precursor for drug development or synthetic drugs
-Pesticide field: can be used for the synthesis of certain pesticides
There are many methods for preparing 3,5-difluororo-2-nitroanisole. The following is a general synthetic route:
First, toluene is reacted with nitric acid to obtain 2-nitrotoluene. Then, 2-nitrotoluene is reacted with hydrogen fluoride under appropriate reaction conditions to obtain 1,5-difluoro-2-nitrotoluene. Finally, 1,5-difluoro-2-nitrotoluene is reacted with methyl ether in the presence of hydrogen iodide to give 3,5-difluoro-2-nitroanisole.
Regarding safety information, 3,5-difluoro-2-nitroanisole is a chemical and needs to be stored and handled correctly. During use, safety procedures must be followed, such as wearing appropriate personal protective equipment (such as gloves, goggles and laboratory coats), avoiding direct contact with the skin and inhaling its vapors. In addition, attention should also be paid to fire and explosion-proof measures to ensure safe use. In the event of an accident, emergency measures shall be taken immediately and professional advice shall be consulted.
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 1,5-Difluoro-3-methoxy-2-nitrobenzene Visit Supplier Webpage Request for quotationCAS: 66684-61-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,5-DIFLUORO-3-METHOXY-2-NITRO-BENZENE Request for quotation
CAS: 66684-61-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 66684-61-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 1-tert-Butyl 2-methyl 5,5-difluoropiperidine-1,2-d icarboxylate... Request for quotation
CAS: 66684-61-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 66684-61-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 1,5-Difluoro-3-methoxy-2-nitrobenzene Visit Supplier Webpage Request for quotationCAS: 66684-61-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,5-DIFLUORO-3-METHOXY-2-NITRO-BENZENE Request for quotation
CAS: 66684-61-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 66684-61-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 1-tert-Butyl 2-methyl 5,5-difluoropiperidine-1,2-d icarboxylate... Request for quotation
CAS: 66684-61-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 66684-61-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
View History


