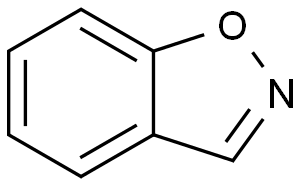
1,2-BENZISOXAZOLE
1,2-BENZISOXAZOLE
CAS: 271-95-4
Molecular Formula: C7H5NO
1,2-BENZISOXAZOLE - Names and Identifiers
| Name | 1,2-BENZISOXAZOLE |
| Synonyms | ISOINDOXAZINE 1,2-benzisozole 4,5-Benzisoazole 1-Oxa-2-azaindene 1,2-BENZISOXAZOLE 4,5-Benzisoxazole 1,2-benzoisoxazole 1-Oxa-2-aza-1H-indene |
| CAS | 271-95-4 |
| EINECS | 205-983-1 |
1,2-BENZISOXAZOLE - Physico-chemical Properties
| Molecular Formula | C7H5NO |
| Molar Mass | 119.12 |
| Density | 1.174 g/mL at 25 °C(lit.) |
| Melting Point | 139 °C |
| Boling Point | 90-92 °C15 mm Hg(lit.) |
| Flash Point | 86°C |
| Water Solubility | Soluble in water. 4.1 mg/mL in water at 25°C |
| Appearance | Shape Oil, color Colourless |
| pKa | -2.03±0.30(Predicted) |
| Storage Condition | 2-8°C |
| Stability | Light Sensitive |
| Refractive Index | n20/D 1.561(lit.) |
| MDL | MFCD00005852 |
| Use | Application 1, 2-benzoxazole can be widely used in organic synthesis. It can be prepared by 2-hydroxybenzaldehyde as the reaction raw material and the intermediate (E)-2-hydroxybenzaldehyde oxime under the action of hydroxylamine hydrochloride, which is further prepared by reacting with triphenylphosphine/DEAD. |
1,2-BENZISOXAZOLE - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 8 |
1,2-BENZISOXAZOLE - Nature
| Solubility | Chloroform (Slightly), Methanol (Slightly) |
| BRN | 2154 |
1,2-BENZISOXAZOLE - Preparation method
1)(E)-Synthesis of 2-hydroxybenzaldehyde oxime Triethylamine (190mmol)
Slowly add to a solution of 2-hydroxybenzaldehyde (164mmol) and hydroxylamine hydrochloride (197mmol) in ethanol (200mL), and heat the reaction mixture at 95°C for 5h. The reaction mixture was concentrated and the residue was extracted with ethyl acetate (2 × 150mL) and water (100mL). Wash the combined organic layer with water (3 × 150mL), dry (magnesium sulfate) and concentrate. The residue was purified by rapid chromatography (1/100 ethyl acetate/petroleum ether) to provide (E)-2-hydroxybenzaldehyde oxime in the form of a white solid with a yield of 43%.
2) Synthesis of 1,2-benzoisazole At 0 ℃, within 4 hours, add DEAD(23.0mmol) tetrahydrofuran (150mL) solution to (E)-2-hydroxybenzaldehyde oxime (21.9mmol) and triphenylphosphine (23.0mmol) tetrahydrofuran (300mL) solution, stir the reaction mixture at 0 ℃ for another 60 minutes and concentrate. The residue was purified by rapid chromatography (1/100 ethyl acetate/petroleum ether) to obtain yellow oily 1,2-benzoisazole in 66% yield.
Supplier List
CAS: 271-95-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 271-95-4
Tel: 86-10-10086 81625225
Email: yeqim@boyechemicals.com
Mobile: 86-15387090420
QQ: 1377812380
Wechat: 15387090420
CAS: 271-95-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 271-95-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 271-95-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 271-95-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 271-95-4
Tel: 86-10-10086 81625225
Email: yeqim@boyechemicals.com
Mobile: 86-15387090420
QQ: 1377812380
Wechat: 15387090420
CAS: 271-95-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 271-95-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 271-95-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
N'-hydroxy-6-(1H-pyrazol-1-yl)pyridine-3-carboximidamide
3-甲氧基-2-溴-6-碘吡啶
1217814-29-3
2,2,5,5-四甲基四氢呋喃
5980-24-5
RAC-米拉贝隆
3,4-dihydro-1(2H)-quinolinyl{4-[3,4-dihydro-1(2H)-quinolinylcarbonyl]cyclohexyl}methanone
N,N'-(Butane-1,4-diyl)bis(3-oxobutanamide)
112339-02-3




