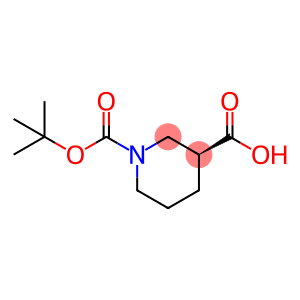
(S)-BOC-哌啶甲酸BOC-(S)-3-甲酸哌啶1-BOC-L-哌啶甲酸
L-1-Boc-Nipecotic acid
CAS: 88495-54-9
Molecular Formula: C11H19NO4
(S)-BOC-哌啶甲酸BOC-(S)-3-甲酸哌啶1-BOC-L-哌啶甲酸 - Names and Identifiers
(S)-BOC-哌啶甲酸BOC-(S)-3-甲酸哌啶1-BOC-L-哌啶甲酸 - Physico-chemical Properties
| Molecular Formula | C11H19NO4 |
| Molar Mass | 229.27 |
| Density | 1.164±0.06 g/cm3(Predicted) |
| Melting Point | 159-162°C(lit.) |
| Boling Point | 353.2±35.0 °C(Predicted) |
| Flash Point | 167.4°C |
| Vapor Presure | 6.15E-06mmHg at 25°C |
| pKa | 4.49±0.20(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.496 |
| MDL | MFCD02179172 |
(S)-BOC-哌啶甲酸BOC-(S)-3-甲酸哌啶1-BOC-L-哌啶甲酸 - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R50 - Very Toxic to aquatic organisms |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3077 9/PG 3 |
| WGK Germany | 3 |
| HS Code | 29333990 |
| Hazard Class | IRRITANT |
(S)-BOC-哌啶甲酸BOC-(S)-3-甲酸哌啶1-BOC-L-哌啶甲酸 - Reference Information
| Introduction | piperidine N-Boc-(S)-3-carboxylate is solid at normal temperature and pressure, it can be used as a pharmaceutical intermediate in the manufacture of local anesthetics, analgesics, fungicides, wetting agents, epoxy resin curing agents, rubber vulcanization accelerators and the like. |
| Use | N-Boc-(S)-3-carboxylic acid piperidine can be used for the preparation of local anesthetics, analgesics, fungicides, wetting agents, etc. In organic synthesis conversion, the carboxyl group in the structure can be reduced to hydroxyl group by borane tetrahydrofuran solution, and the carboxyl group can also be converted to Ester group or amide group. |
| preparation method | triethylamine (1.92 ML, 13.66 mmol, 1.2 equiv) was added at 0°C. And Boc2O(3.48 ML, 15.93 mmol, 1.4 equiv) was added to a solution of nipaginic acid (1.5g, 11.38 mmol, 1 equiv) in methanol (44 ml), the resulting reaction mixture was warmed to room temperature and the mixture was stirred at room temperature overnight. To the mixture was added a mixed solution of dichloromethane and water = 5:1(60 ml), and then hydrochloric acid (1N) was added to adjust the pH of the reaction system to pH = 3. The aqueous phase was extracted with dichloromethane, the combined organic phases were dried over anhydrous magnesium sulfate, the mixture was filtered to remove magnesium sulfate solids, and the filtrate was concentrated under reduced pressure. The residue was separated and purified by silica gel column chromatography (dichloromethane to methanol 95:5 to 85:15) to give piperidine N-Boc-(S)-3-carboxylate. Figure N-Boc-(S)-piperidine -3-carboxylate synthesis |
Last Update:2024-04-09 15:16:53
Supplier List
Featured ProductsSpot supply
Product Name: L-1-Boc-Nipecotic acid Request for quotationCAS: 88495-54-9
Tel: +86-18980427282
Email: hk999@hengkangtech.com
Mobile: +86-18980427282
QQ: 1525088774
Wechat: 18980427282
WhatsApp: 15008222507
Linkedin: linkedin.com/in/teresa-li-806a0095
Product List: View Catalog
Spot supply
Product Name: L-1-Boc-Nipecotic acid Visit Supplier Webpage Request for quotationCAS: 88495-54-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: L-1-Boc-Nipecotic acid Request for quotation
CAS: 88495-54-9
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
CAS: 88495-54-9
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
Spot supply
Product Name: L-1-Boc-Nipecotic acid Request for quotationCAS: 88495-54-9
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Spot supply
Product Name: S-1-Boc-piperidine-3-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 88495-54-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-(+)-Glycidyl Phthalimide Request for quotation
CAS: 88495-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 88495-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: L-1-Boc-Nipecotic acid Request for quotation
CAS: 88495-54-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 88495-54-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: (S)-1-Boc-piperidine-3-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 88495-54-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured ProductsSpot supply
Product Name: L-1-Boc-Nipecotic acid Request for quotationCAS: 88495-54-9
Tel: +86-18980427282
Email: hk999@hengkangtech.com
Mobile: +86-18980427282
QQ: 1525088774
Wechat: 18980427282
WhatsApp: 15008222507
Linkedin: linkedin.com/in/teresa-li-806a0095
Product List: View Catalog
Spot supply
Product Name: L-1-Boc-Nipecotic acid Visit Supplier Webpage Request for quotationCAS: 88495-54-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: L-1-Boc-Nipecotic acid Request for quotation
CAS: 88495-54-9
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
CAS: 88495-54-9
Tel: 17505222756
Email: pules.cn@gmail.com
Mobile: +86-17551318830
Spot supply
Product Name: L-1-Boc-Nipecotic acid Request for quotationCAS: 88495-54-9
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Spot supply
Product Name: S-1-Boc-piperidine-3-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 88495-54-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-(+)-Glycidyl Phthalimide Request for quotation
CAS: 88495-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 88495-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: L-1-Boc-Nipecotic acid Request for quotation
CAS: 88495-54-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 88495-54-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: (S)-1-Boc-piperidine-3-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 88495-54-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History







