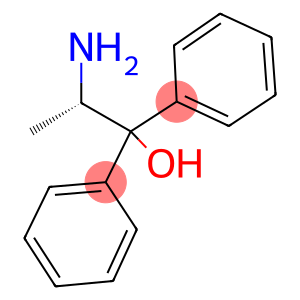
(S)-(−)-2-Amino-1,1-diphenyl-1-propanol
(S)-(−)-2-Amino-1,1-diphenyl-1-propanol
CAS: 78603-91-5
Molecular Formula: C15H17NO
(S)-(−)-2-Amino-1,1-diphenyl-1-propanol - Names and Identifiers
(S)-(−)-2-Amino-1,1-diphenyl-1-propanol - Physico-chemical Properties
| Molecular Formula | C15H17NO |
| Molar Mass | 227.3 |
| Density | 1.112±0.06 g/cm3(Predicted) |
| Melting Point | 100-102°C(lit.) |
| Boling Point | 407.8±40.0 °C(Predicted) |
| Specific Rotation(α) | -90 º (C=1 IN CHLOROFORM) |
| BRN | 3052863 |
| pKa | 11.25±0.50(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| MDL | MFCD00145253 |
(S)-(−)-2-Amino-1,1-diphenyl-1-propanol - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 9 |
(S)-(−)-2-Amino-1,1-diphenyl-1-propanol - Reference Information
| Application prospects | In asymmetric synthesis, the synthesis of chiral catalysts or chiral additives is very important. Chiral amino alcohols are widely used to catalyze asymmetric synthesis The ligand or catalyst of the reaction is also an important precursor for the synthesis of many chiral drugs, natural products and chemical and biological functional materials. (S)-2-amino -1, 1-diphenyl-1-propanol belongs to the optically active β-amino alcohol. Asymmetric reduction of prochiral ketones, asymmetric catalytic epoxidation, asymmetric catalytic alkylation of carbonyl compounds, etc. (S)-2-amino -1, 1-diphenyl-1-propanol has also been reported as an intermediate in the synthesis of chiral complexes in asymmetric cyclopropanation. |
| Preparation | The related synthesis methods reported in the literature are divided into two categories: one is L-alanine, L-alanine hydrochloride or L-alanine methyl ester directly react with phenyl magnesium bromide to prepare (S)-2-amino -1, 1-diphenyl -1-propanol, its shortcomings are complex products and difficult purification, and the yield is low; the other type is prepared by 4-step reaction with L-alanine as the starting material, that is, the L-2-benzylcarbonylaminalanine methyl ester prepared by methyl esterification and benzyloxycarbonyl protection React with phenyl magnesium bromide to obtain (S)-benzyloxycarbonylamino -1, 1-diphenyl-1-propanol, the latter catalyzes the hydrogenation of debenzyloxycarbonyl to obtain (S)-2-amino -1, 1-diphenyl-1-propanol. This paper reports a new method for the synthesis of (S)-2-amino -1, 1-diphenyl -1-propanol. In the presence of potassium carbonate, using water-ethanol as a mixed solvent, the amino and carboxyl groups in the L-alanine molecule react with benzyl chloride to form N,N-dibenzyl-L-alanine benzyl ester (1);1 and phenylmagnesium bromide by Grignard reaction to obtain 1, 1-diphenyl-2-(N,N-dibenzylamino)-1-propanol (2);2 After Pd/C catalytic hydrogenation debenzyl protection 3, the total yield is 48-1%, the intermediate product and the target product of the synthesis method are easy to separate and purify. (S)-2-amino -1, 1-diphenyl-1-propanol synthesis reaction formula is as follows: |
| Uses | (S)-2-amino-1, 1-diphenyl-1-propanol is an alcohol derivative that can be used as an intermediate in organic synthesis. |
Last Update:2024-04-09 20:45:29
Supplier List
Spot supply
Product Name: (S)-(-)-2-Amino-1,1-diphenyl-1-propanol Visit Supplier Webpage Request for quotationCAS: 78603-91-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-Amino-naphthalen-1-yl-acetic acid hydrochloride Request for quotation
CAS: 78603-91-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 78603-91-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Multiple Specifications
Product Name: (S)-(-)-2-AMINO-1,1-DIPHENYL-1-PROPANOL Request for quotationCAS: 78603-91-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (S)-(−)-2-Amino-1,1-diphenyl-1-propanol Visit Supplier Webpage Request for quotation
CAS: 78603-91-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 78603-91-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-(-)-2-Amino-1,1-diphenyl-1-propanol Visit Supplier Webpage Request for quotationCAS: 78603-91-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-Amino-naphthalen-1-yl-acetic acid hydrochloride Request for quotation
CAS: 78603-91-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 78603-91-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Multiple Specifications
Product Name: (S)-(-)-2-AMINO-1,1-DIPHENYL-1-PROPANOL Request for quotationCAS: 78603-91-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (S)-(−)-2-Amino-1,1-diphenyl-1-propanol Visit Supplier Webpage Request for quotation
CAS: 78603-91-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 78603-91-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



