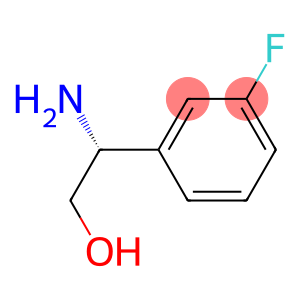
(R)-2-Amino-2-(3-fluorophenyl)ethanol
(2R)-2-Amino-2-(3-fluoropheny)ethanol
CAS: 1213968-04-7
Molecular Formula: C8H10FNO
(R)-2-Amino-2-(3-fluorophenyl)ethanol - Names and Identifiers
(R)-2-Amino-2-(3-fluorophenyl)ethanol - Physico-chemical Properties
| Molecular Formula | C8H10FNO |
| Molar Mass | 155.17 |
| Density | 1.208±0.06 g/cm3(Predicted) |
| Boling Point | 286.3±25.0 °C(Predicted) |
| pKa | 12.40±0.10(Predicted) |
| Storage Condition | 2-8℃ |
(R)-2-Amino-2-(3-fluorophenyl)ethanol - Introduction
(R)-2-amino -2-(3-fluorophenyl) ethanol, also known as (R)-3-fluorophenylethylamine alcohol, is a chiral molecule, chemical formula is C8H10FNO. The following is a description of its nature, use, formulation and safety information:
Nature:
(R)-2-Amino -2-(3-fluorophenyl) ethanol is a colorless liquid or solid with a sweet taste. It is a chiral molecule and exists in two enantiomers, (R) and (S). Typically, only (R)-2-amino -2-(3-fluorophenyl) ethanol is present.
Use:
(R)-2-amino -2-(3-fluorophenyl) ethanol is an important intermediate in organic synthesis. It can be used in the preparation of pharmaceuticals, pesticides and specialty chemicals. This compound is commonly used as a chiral ligand and participates in a variety of organic synthesis reactions, such as asymmetric catalytic reactions.
Preparation Method:
There are many ways to prepare (R)-2-amino -2-(3-fluorophenyl) ethanol, the most common of which is through the reduction of 2-amino -2-(3-fluorophenyl) acetonitrile. Phosphorous acid or ammonium phosphite is usually used as the reducing agent.
Safety Information:
(R)-2-amino -2-(3-fluorophenyl) ethanol is less toxic, but as any chemical substance, you must pay attention to the correct use and storage. It should be stored in a dry, cool, well-ventilated place, away from fire and oxidants. During handling, direct contact with skin, eyes and mucous membranes should be avoided. In case of accidental contact, rinse immediately with plenty of water and seek medical help.
It is important to note that since it is a chiral molecule and there are two enantiomers, special attention needs to be paid to the selectivity of the desired enantiomer (R) or (S) when used to ensure the chiral purity and effect of the desired synthetic target.
Nature:
(R)-2-Amino -2-(3-fluorophenyl) ethanol is a colorless liquid or solid with a sweet taste. It is a chiral molecule and exists in two enantiomers, (R) and (S). Typically, only (R)-2-amino -2-(3-fluorophenyl) ethanol is present.
Use:
(R)-2-amino -2-(3-fluorophenyl) ethanol is an important intermediate in organic synthesis. It can be used in the preparation of pharmaceuticals, pesticides and specialty chemicals. This compound is commonly used as a chiral ligand and participates in a variety of organic synthesis reactions, such as asymmetric catalytic reactions.
Preparation Method:
There are many ways to prepare (R)-2-amino -2-(3-fluorophenyl) ethanol, the most common of which is through the reduction of 2-amino -2-(3-fluorophenyl) acetonitrile. Phosphorous acid or ammonium phosphite is usually used as the reducing agent.
Safety Information:
(R)-2-amino -2-(3-fluorophenyl) ethanol is less toxic, but as any chemical substance, you must pay attention to the correct use and storage. It should be stored in a dry, cool, well-ventilated place, away from fire and oxidants. During handling, direct contact with skin, eyes and mucous membranes should be avoided. In case of accidental contact, rinse immediately with plenty of water and seek medical help.
It is important to note that since it is a chiral molecule and there are two enantiomers, special attention needs to be paid to the selectivity of the desired enantiomer (R) or (S) when used to ensure the chiral purity and effect of the desired synthetic target.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: (R)-b-AMino-3-fluoro-benzeneethanol Visit Supplier Webpage Request for quotationCAS: 1213968-04-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (R)-b-AMino-3-fluoro-benzeneethanol Visit Supplier Webpage Request for quotation
CAS: 1213968-04-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1213968-04-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (R)-b-AMino-3-fluoro-benzeneethanol Visit Supplier Webpage Request for quotationCAS: 1213968-04-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (R)-b-AMino-3-fluoro-benzeneethanol Visit Supplier Webpage Request for quotation
CAS: 1213968-04-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1213968-04-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

