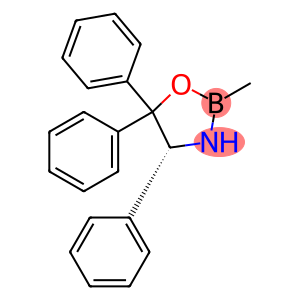
(4R)-2-Methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine
(r)-b-methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine
CAS: 155268-88-5
Molecular Formula: C21H20BNO
(4R)-2-Methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine - Names and Identifiers
(4R)-2-Methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine - Physico-chemical Properties
| Molecular Formula | C21H20BNO |
| Molar Mass | 313.2 |
| Density | 1.13 |
(4R)-2-Methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine - Introduction
Nature:
(r)-b-methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine is a white to light yellow solid. It has a high melting point and boiling point, and is stable at room temperature. It is poorly soluble in solvents but readily soluble in organic solvents such as chloroform, dichloromethane and dimethylformamide.
Use:
(r)-b-methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine has important applications in organic synthesis. It can be used as catalyst and ligand, and has good catalytic activity in enantioselective synthesis. In addition, it is also used in halogenation reactions and substitution reactions in organic synthesis.
Method:
(r)-b-methyl-4, the synthesis of 5,5-triphenyl-1,3,2-oxazaborolidine is usually carried out by chemical synthesis. A common method is to add benzyl lithium to oxazole formaldehyde to obtain dibenzyl oxazole alcohol, and then react with triphenylboronic acid ester to finally obtain the target product.
Safety Information:
(r)-b-methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine is relatively stable under general conditions of use. However, it can be irritating to the eyes, skin and respiratory system. Therefore, appropriate protective equipment such as gloves and glasses should be worn during use. In addition, during storage and handling, avoid contact with oxidants and strong acids to avoid danger. Before use, you should carefully read and follow the relevant safety instructions.
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: (4R)-2-Methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine Request for quotation
CAS: 155268-88-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 155268-88-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: (4R)-2-Methyl-4,5,5-triphenyl-1,3,2-oxazaborolidine Request for quotation
CAS: 155268-88-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 155268-88-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
