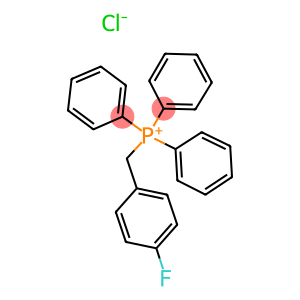
(4-fluorophenyl)methyl-triphenylphosphanium
(4-Fluorobenzyl)triphenylphosphonium chloride
CAS: 3462-95-1
Molecular Formula: C25H21ClFP
(4-fluorophenyl)methyl-triphenylphosphanium - Names and Identifiers
| Name | (4-Fluorobenzyl)triphenylphosphonium chloride |
| Synonyms | (4-fluorophenyl)methyl-triphenylphosphanium (4-FLUOROBENZYL)TRIPHENYLPHOSPHONIUM CHLORIDE (4-Fluorobenzyl)triphenylphosphonium chloride (4-Fluorobenzyl)(triphenyl)phosphonium chloride (4-Fluorophenylmethyl)triphenylphosphoniumchloride (4-fluorophenyl)methyl-triphenylphosphanium,chloride |
| CAS | 3462-95-1 |
| InChI | InChI=1/C25H21FP.ClH/c26-22-18-16-21(17-19-22)20-27(23-10-4-1-5-11-23,24-12-6-2-7-13-24)25-14-8-3-9-15-25;/h1-19H,20H2;1H/q+1;/p-1 |
(4-fluorophenyl)methyl-triphenylphosphanium - Physico-chemical Properties
| Molecular Formula | C25H21ClFP |
| Molar Mass | 406.86 |
| Melting Point | 310-312°C |
| BRN | 4075331 |
| Storage Condition | Inert atmosphere,Room Temperature |
| Sensitive | Hygroscopic |
| MDL | MFCD00031637 |
(4-fluorophenyl)methyl-triphenylphosphanium - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
(4-fluorophenyl)methyl-triphenylphosphanium - Introduction
(4-Fluorobenzyl)triphenylphosphonium chloride is an organic compound with a chemical formula of C21H17ClFP and a molecular weight of 358.80g/mol. The following is a detailed description of its nature, use, formulation and safety information:
Nature:
(4-Fluorobenzyl)triphenylphosphonium chloride is a white crystalline solid, soluble in non-polar solvents such as diethyl ether and chloroform. It is stable in air, but decomposes in humid conditions. This compound is susceptible to light and thermal decomposition.
Use:
(4-Fluorobenzyl)triphenylphosphonium chloride are commonly used as reagents in organic synthesis. It can serve as a source of low coordinating anions, such as fluoride, chloride and nitrate ions. In addition, it can also be used in the synthesis of fluorescent dyes, organometallic chemistry and catalytic reactions.
Preparation Method:
(4-Fluorobenzyl)triphenylphosphonium chloride can be prepared by the reaction of phenylphosphine chloride and 4-fluorobenzyl bromide. First, phenylphosphine chloride is dissolved in an organic solvent, and then 4-fluorobenzyl bromide is added for reaction to form the product. Finally, the pure product is obtained by crystallization and purification.
Safety Information:
(4-Fluorobenzyl)triphenylphosphonium chloride can be used safely under normal operating conditions, the following matters still need to be paid attention to: avoid contact with skin and eyes, and wear appropriate personal protective equipment, such as laboratory gloves and goggles. In addition, it should be operated under well-ventilated conditions to avoid inhaling its dust or vapor mist. If ingested or inhaled by mistake, seek medical help immediately.
Nature:
(4-Fluorobenzyl)triphenylphosphonium chloride is a white crystalline solid, soluble in non-polar solvents such as diethyl ether and chloroform. It is stable in air, but decomposes in humid conditions. This compound is susceptible to light and thermal decomposition.
Use:
(4-Fluorobenzyl)triphenylphosphonium chloride are commonly used as reagents in organic synthesis. It can serve as a source of low coordinating anions, such as fluoride, chloride and nitrate ions. In addition, it can also be used in the synthesis of fluorescent dyes, organometallic chemistry and catalytic reactions.
Preparation Method:
(4-Fluorobenzyl)triphenylphosphonium chloride can be prepared by the reaction of phenylphosphine chloride and 4-fluorobenzyl bromide. First, phenylphosphine chloride is dissolved in an organic solvent, and then 4-fluorobenzyl bromide is added for reaction to form the product. Finally, the pure product is obtained by crystallization and purification.
Safety Information:
(4-Fluorobenzyl)triphenylphosphonium chloride can be used safely under normal operating conditions, the following matters still need to be paid attention to: avoid contact with skin and eyes, and wear appropriate personal protective equipment, such as laboratory gloves and goggles. In addition, it should be operated under well-ventilated conditions to avoid inhaling its dust or vapor mist. If ingested or inhaled by mistake, seek medical help immediately.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: (4-Fluorobenzyl)triphenylphosphonium chloride Visit Supplier Webpage Request for quotationCAS: 3462-95-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (4-Fluorotetrahydro-2H-pyran-4-yl)MethanaMine hydrochloride Request for quotation
CAS: 3462-95-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 3462-95-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (4-FLUOROBENZYL)TRIPHENYLPHOSPHONIUM CHLORIDE Request for quotation
CAS: 3462-95-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3462-95-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: (4-Fluorobenzyl)Triphenylphosphonium Chloride Visit Supplier Webpage Request for quotationCAS: 3462-95-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (4-Fluorobenzyl)triphenylphosphonium chloride Visit Supplier Webpage Request for quotationCAS: 3462-95-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (4-Fluorotetrahydro-2H-pyran-4-yl)MethanaMine hydrochloride Request for quotation
CAS: 3462-95-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 3462-95-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (4-FLUOROBENZYL)TRIPHENYLPHOSPHONIUM CHLORIDE Request for quotation
CAS: 3462-95-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3462-95-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: (4-Fluorobenzyl)Triphenylphosphonium Chloride Visit Supplier Webpage Request for quotationCAS: 3462-95-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



