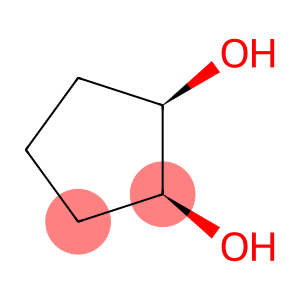
(1S)-Cyclopentane-1α,2α-diol
(1R,2S)-cyclopentane-1,2-diol
CAS: 5057-98-7
Molecular Formula: C5H10O2
(1S)-Cyclopentane-1α,2α-diol - Names and Identifiers
| Name | (1R,2S)-cyclopentane-1,2-diol |
| Synonyms | TIMTEC-BB SBB008502 CIS-1,2-CYCLOPENTANEDIOL CIS-CYCLOPENTANE-1,2-DIOL 1,2-Cyclopentanediol, cis- (1S)-Cyclopentane-1α,2α-diol (1R,2S)-1,2-Cyclopentanediol CIS-1,2-DIHYDROXYCYCLOPENTANE (1R,2S)-cyclopentane-1,2-diol |
| CAS | 5057-98-7 |
| InChI | InChI=1/C5H10O2/c6-4-2-1-3-5(4)7/h4-7H,1-3H2/t4-,5+ |
(1S)-Cyclopentane-1α,2α-diol - Physico-chemical Properties
| Molecular Formula | C5H10O2 |
| Molar Mass | 102.13 |
| Density | 1.0042 (rough estimate) |
| Melting Point | 24-28°C(lit.) |
| Boling Point | 108-109°C20mm Hg(lit.) |
| Flash Point | >230°F |
| Vapor Presure | 0.0304mmHg at 25°C |
| Appearance | Liquid After Melting |
| Color | Clear slightly yellow |
| BRN | 1901117 |
| pKa | 14.48±0.40(Predicted) |
| Storage Condition | Sealed in dry,2-8°C |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.477-1.479 |
(1S)-Cyclopentane-1α,2α-diol - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 3-10 |
| HS Code | 29061900 |
(1S)-Cyclopentane-1α,2α-diol - Introduction
(1R,2S)-cyclopentane-1,2-diol is an organic compound with the chemical formula C5H10O2. Its structure contains a five-membered ring and two hydroxyl functional groups.
Nature:
(1R,2S)-cyclopentane-1,2-diol is a colorless or white crystalline solid, soluble in water and some organic solvents. It is a chiral molecule with two possible stereoisomers: cis (1R,2S) and trans (1S,2S). (1R,2S)-cyclopentane-1,2-diol often exists as a cis isomer. It has a melting point of about 65-67°C and a boiling point of about 268-271°C.
Use:
(1R,2S)-cyclopentane-1,2-diol has a wide range of applications in the chemical and pharmaceutical industries. It is an important synthetic intermediate, which is often used in the preparation of other compounds in organic synthesis reactions, such as drugs, fragrances, dyes and polymer materials.
Preparation Method:
(1R,2S)-cyclopentane-1,2-diol can be prepared in a variety of ways. The common method is obtained by the hydrolysis of cyclopentanedione, which reacts with water under acidic conditions to give (1R,2S)-cyclopentane-1,2-diol. It can also be obtained by oxidation of cis-2-buten-1-ol.
Safety Information:
(1R,2S)-cyclopentane-1,2-diol is generally considered to be a relatively safe compound, but it is still necessary to pay attention to the safety of its use and storage. In the process of operation, it should avoid contact with skin, eyes and mucous membranes. During storage, avoid high temperature and contact with harmful substances such as oxidants, acids and alkalis.
it should be noted that the information mentioned above is for reference only, and the specific nature, use, method and safety information should also be comprehensively understood in combination with the actual situation and relevant literature.
Nature:
(1R,2S)-cyclopentane-1,2-diol is a colorless or white crystalline solid, soluble in water and some organic solvents. It is a chiral molecule with two possible stereoisomers: cis (1R,2S) and trans (1S,2S). (1R,2S)-cyclopentane-1,2-diol often exists as a cis isomer. It has a melting point of about 65-67°C and a boiling point of about 268-271°C.
Use:
(1R,2S)-cyclopentane-1,2-diol has a wide range of applications in the chemical and pharmaceutical industries. It is an important synthetic intermediate, which is often used in the preparation of other compounds in organic synthesis reactions, such as drugs, fragrances, dyes and polymer materials.
Preparation Method:
(1R,2S)-cyclopentane-1,2-diol can be prepared in a variety of ways. The common method is obtained by the hydrolysis of cyclopentanedione, which reacts with water under acidic conditions to give (1R,2S)-cyclopentane-1,2-diol. It can also be obtained by oxidation of cis-2-buten-1-ol.
Safety Information:
(1R,2S)-cyclopentane-1,2-diol is generally considered to be a relatively safe compound, but it is still necessary to pay attention to the safety of its use and storage. In the process of operation, it should avoid contact with skin, eyes and mucous membranes. During storage, avoid high temperature and contact with harmful substances such as oxidants, acids and alkalis.
it should be noted that the information mentioned above is for reference only, and the specific nature, use, method and safety information should also be comprehensively understood in combination with the actual situation and relevant literature.
Last Update:2024-04-09 15:17:58
Supplier List
Spot supply
Product Name: cis-1,2-Cyclopentanediol 98% Visit Supplier Webpage Request for quotationCAS: 5057-98-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CIS-1,2-CYCLOPENTANEDIOL Request for quotation
CAS: 5057-98-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5057-98-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Cis-1,2-Cyclopentanediol Visit Supplier Webpage Request for quotation
CAS: 5057-98-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5057-98-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: cis-1,2-Cyclopentanediol 98% Visit Supplier Webpage Request for quotationCAS: 5057-98-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CIS-1,2-CYCLOPENTANEDIOL Request for quotation
CAS: 5057-98-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5057-98-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Cis-1,2-Cyclopentanediol Visit Supplier Webpage Request for quotation
CAS: 5057-98-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5057-98-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


