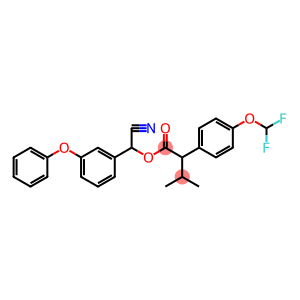
(+-)-cyano(3-phenoxyphenyl)methyl(+)-4-(difluoromethoxy)-alpha-(1-methylethy
Flucythrinate
CAS: 70124-77-5
Molecular Formula: C26H23F2NO4
(+-)-cyano(3-phenoxyphenyl)methyl(+)-4-(difluoromethoxy)-alpha-(1-methylethy - Names and Identifiers
(+-)-cyano(3-phenoxyphenyl)methyl(+)-4-(difluoromethoxy)-alpha-(1-methylethy - Physico-chemical Properties
| Molecular Formula | C26H23F2NO4 |
| Molar Mass | 451.47 |
| Density | 1.189 g/cm3 (22℃) |
| Boling Point | 108℃ (0.35mmHg) |
| Flash Point | -18°C |
| Water Solubility | 0.5 mg l-1 (22 °C) |
| Vapor Presure | 1.2×10-6 Pa (25 °C) |
| Appearance | neat |
| Specific Gravity | 1.189 (22℃) |
| BRN | 2195795 |
| Storage Condition | 2-8°C |
| Refractive Index | 1.541 (589.3 nm 25℃) |
| Physical and Chemical Properties | The pure product is Amber viscous liquid. B. p.108 °c/46.66, relative density 1.189 (22 °c), vapor pressure 3.2 x 10-5Pa (45 °c). Solubility: acetone> 82%, propanol> 78%, hexane 9%, xylene 181%; Almost insoluble in water (65mg/L), the partition coefficient is 120, at pH 3, hydrolysis half-life of about 40d; PH value 6 is 52d; PH value 9 is 6.3d (both 27 degrees C). |
| Use | Efficient, broad-spectrum, fast, low residue, long residual effect of pyrethroid insecticides, and acaricidal, tick-killing activity. This product is mainly contact and stomach poison, no inhalation effect. Can control tea, tea geometrid, tea moth, cotton bollworm, pink bollworm, the second and third generation of egg incubation period and young insects into the bud, before the bell, citrus leaf moth in the early stage of the shoot or egg incubation period, cabbage, diamondback moth, Spodoptera litura, beet armyworm 3 instar larvae before the onset of use. The drug is easy to produce cross resistance with other pyrethroid pesticides. |
(+-)-cyano(3-phenoxyphenyl)methyl(+)-4-(difluoromethoxy)-alpha-(1-methylethy - Risk and Safety
| Risk Codes | R20/21 - Harmful by inhalation and in contact with skin. R25 - Toxic if swallowed R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R67 - Vapors may cause drowsiness and dizziness R65 - Harmful: May cause lung damage if swallowed R38 - Irritating to the skin R11 - Highly Flammable |
| Safety Description | S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S62 - If swallowed, do not induce vomitting; seek medical advice immediately and show this container or label. |
| UN IDs | UN 2810 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | CY1578620 |
| HS Code | 29269090 |
| Toxicity | LC50 (96-hour) for rainbow trout 0.32 μg/L, bluegill sun?sh 0.71 μg/L, channel cat?sh 0.51 μg/L, sheepshead minnow 1.6 μg/L (Hartley and Kidd, 1987), estuarine mysid 0.008 μg/L, pink shrimp 0.22 μg/L and sheepshead minnow 1.1 μg/L (Schimmel et al., 1983); acute oral LD50 for male and female rats is 81 and 67 mg/kg, respectively (Hartley and Kidd, 1987). |
(+-)-cyano(3-phenoxyphenyl)methyl(+)-4-(difluoromethoxy)-alpha-(1-methylethy - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| toxicity | acute oral LD50 of male rats is 81 mg/kg, female is 67 mg/kg; Acute inhalation of rats is LD50 4.85mg/L; Rabbit acute percutaneous LD50>1000 mg/kg. It is irritating to eyes and skin. The feed containing 300mg/kg of the original drug was fed to the dog for 90 days, and the dog's food intake and body weight decreased, but no macroscopic and microscopic lesions were found. The chronic oral non-effect dose in rats is 60mg/kg per day. No teratogenic, mutagenic and carcinogenic effects were found in animal experiments. The LC50 of carp is 0.01mg/L (96h), the acute oral LD50 of quail is 2708mg/kg, and the LD50 of bee is 0.078 μg/animal. |
| use | pyrethroid insecticides with high efficiency, broad spectrum, fast, low residue and long residual effect, with both acaricidal and tickicidal activities. This product is mainly contact and stomach poison, and has no systemic effect. It can prevent tea caterpillars, tea geometrid, tea moth, cotton bollworm, red bollworm, second and third generations of egg incubation period and before the young insects enter the buds and bolls, citrus leaf miner is in the early stage of shoot or egg incubation period, Cabbage worm, diamondback moth, Spodoptera litura, Spodoptera litura, Spodoptera litura, Spodoptera beet Spodoptera are used before the 3th instar larvae are in full bloom. The drug is prone to cross-resistance with other pyrethroid pesticides. This product is a new variety of fluorine-containing pyrethroids, and its chemical structure is similar to that of fenvalerate. Because this product contains fluorine, it has high efficiency, is more stable to light and heat, and can also control mites and ticks. It is mainly used to control many pests such as Lepidoptera, Diptera, and Coleoptera. It is also used to control cotton bollworms, red bollworms, red spiders, whiteflies, vegetables and fruit trees in cotton fields. |
| production method | using anisaldehyde as the starting material, after reduction, chlorination, cyanoization, isopropylation, hydrolysis, difluoromethyl ether chloride, Esterification and other steps can obtain cyhalothrin. This synthesis route is lengthy, the reaction yield of several steps is low, the source of raw materials is difficult, and the cost is high. Pyrethroid insecticides, especially cyanide valerate (fenvalerate), use the intermediate 2-(p-chlorophenyl)-3-methylbutyric acid to directly hydrolyze to 2-(p-Hydroxyphenyl)-3-methylbutyric acid, and then difluoromethyl ether, acyl chlorination, acyl chlorination, condensation and other reactions to obtain cyfluthrin. This synthetic route has a higher yield. preparation method 1 is synthesized by 2-p-methoxyphenyl isovaleric acid by resolution (invalid racemate), hydrolysis, trifluoromethylation, acyl chlorination and esterification. Including racemization and recovery, the total yield can reach more than 75%. Resolution of 2-p-methoxyphenyl isovaleric acid 27g 2-p-methoxyphenyl isovaleric acid, 15g (-)2-phenethylamine, 1500mL 60% ethanol, dissolved by heating, cooled and recrystallized to obtain 14.5g white needle crystal; The mother liquor was combined, ethanol was evaporated, and 3.5g white crystal was obtained by standing, filtering and recrystallization. A total of 8g of solid was obtained by merging the two crystals. Add 200mL of water and 10mL of concentrated hydrochloric acid. Heat and cool to obtain 11.5g of white solid, m.p. 136~138 ℃, [α]20D = +52.2o, 42.6% yield and 99.24% optical purity. Preparation of (S)(+)-2-p-hydroxyphenylisovaleric acid 10g of the previous product, 40mL of 48% hydrobromic acid and 2mL of glacial acetic acid were added. Heating at 126 ℃ ~ 130 ℃, reflux reaction for 3h, cooling, water recrystallization to obtain 9g white needle crystal, m.p.189 ~ 191 ℃,[α]20D = +52.4o, yield 96.5%. Preparation of (S)(+)-2-p-difluoromethoxy phenyl isovaleric acid 10g of the previous product is added with 19g of sodium hydroxide, 30mL of water and 60mL of dioxane, heated to 80 ℃, passed with fluoropexia -22 about 40g, cooled, poured into 250mL of water, washed with ether, acidified with concentrated hydrochloric acid, extracted with ether, dried with anhydrous sodium sulfate, distilled with ether to obtain light brown mass, and 1:1 n-hexane-dichloromethane extraction, residue white powder 5.1g,m p 189~191 ℃,[α]20D = +2.4o. The extract was evaporated under vacuum to obtain light orange semi-solid 5.5g,[α]20D = +40.2o, with 3.7% yield and 99.5% optical purity. Preparation of (S)-2-p-difluoromethoxyphenyl isovaleroyl chloride 1.7g([α]20D = +40.2o), 25mL toluene, 1mL sulfoxide chloride and 1 drop of dimethylformamide were added to the previous product, heated and refluxed for 0.5h, cooled, and evaporated toluene under reduced pressure to obtain 1.9g of orange viscous solution, [α]20D = +37.4o. $4 synthesis of fenvalerate 1.31g of m-phenoxybenzaldehyde, 0.2g of tetrabutylammonium bromide, 25mL of toluene, 1.5g of potassium cyanide and 3mL of water are stirred and added dropwise (S)(+)-2-p-difluoromethoxyphenyl isopentyl chloride-toluene mixed solution 10mL (containing 1.9g of acid chloride) at normal temperature, stirred for 1h after dripping, and 10mL of water is added, wash once with 10% Na2CO3, water, 10% HCl and saturated salt water 10mL respectively, dry with anhydrous sodium sulfate, and evaporate toluene under reduced pressure to obtain 3.7g of dark red viscous liquid. Silica gel column chromatography was used for separation, about 10mL of light yellow chromatography was collected, and solvent was distilled to obtain 3.1g of light green viscous liquid, 19D = 1.5460,[α]20D =-5.43o, with 99% yield and 98.5% optical purity. The racemization reaction of (R)-2-p-methoxyphenylisovaleric acid, the (R)-2-p-methoxyisovaleric acid generated by the reaction is racemized (the yield can reach 98.5%) to improve the total yield of this synthetic route. Preparation method 2 Preparation method 3 |
| category | pesticide |
| toxicity classification | highly toxic |
| acute toxicity | oral-rat LD50: 67 mg/kg; Oral-mouse LD50: 76 mg/kg |
| flammability hazard characteristics | flammable; combustion produces toxic nitrogen oxides and fluoride gases |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from food raw materials storage and transportation |
| fire extinguishing agent | dry powder, foam, sand |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:04
Supplier List
Spot supply
Product Name: Flucythrinate solution Visit Supplier Webpage Request for quotationCAS: 70124-77-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: FLUCYTHRINATE Request for quotation
CAS: 70124-77-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 70124-77-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple Specifications
Product Name: Flucythrinate Visit Supplier Webpage Request for quotationCAS: 70124-77-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Flucythrinate solution Visit Supplier Webpage Request for quotationCAS: 70124-77-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: FLUCYTHRINATE Request for quotation
CAS: 70124-77-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 70124-77-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple Specifications
Product Name: Flucythrinate Visit Supplier Webpage Request for quotationCAS: 70124-77-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


