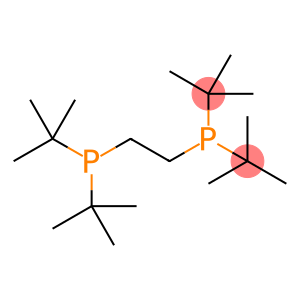
2-(tert-butyl(2-(di-tert-butylphosphino)ethyl)phosphino)-2-methylpropane
Phosphine, 1,2-ethanediylbis[bis(1,1-dimethylethyl)-
CAS: 107783-62-0
Molecular Formula: C18 H40 P2
2-(tert-butyl(2-(di-tert-butylphosphino)ethyl)phosphino)-2-methylpropane - Names and Identifiers
| Name | Phosphine, 1,2-ethanediylbis[bis(1,1-dimethylethyl)- |
| Synonyms | 1,2-Bis(di-tert-butylphosphino)ethane 1,2-BIS(DI-TERT-BUTYLPHOSPHINO)ETHANE Phosphine, 1,2-ethanediylbis[bis(1,1-dimethylethyl)- Phosphine, 1,1'-(1,2-ethanediyl)bis[1,1-bis(1,1-dimethylethyl)- 2-(tert-butyl(2-(di-tert-butylphosphino)ethyl)phosphino)-2-methylpropane |
| CAS | 107783-62-0 |
2-(tert-butyl(2-(di-tert-butylphosphino)ethyl)phosphino)-2-methylpropane - Physico-chemical Properties
| Molecular Formula | C18 H40 P2 |
| Molar Mass | 318.46 |
| Boling Point | 361.2±25.0 °C(Predicted) |
| Storage Condition | -20℃ |
2-(tert-butyl(2-(di-tert-butylphosphino)ethyl)phosphino)-2-methylpropane - Introduction
1,2-bis (di-tert-butylphosphino) ethane, also known as 1,2-bisphosphine, is an organic compound. Its chemical formula is C10H24P2 and its molecular weight is 208.27g/mol.
Nature:
1,2-Bis (di-tert-butylphosphino) ethane is a colorless liquid with a special smell. It is soluble in common organic solvents such as ethanol, acetone and dimethylformamide. It is a non-volatile liquid at room temperature and has a relatively low vapor pressure.
Use:
1,2-Bis (di-tert-butylphosphine) ethane is an important organic synthesis reagent, which is widely used in organic synthesis reactions. It can be used as a ligand in the catalyst system for hydrogenation, rearrangement, selective reduction and other reactions in organic synthesis. It can also be used in the synthesis of phosphinone compounds and transition metal phosphine complexes.
Preparation Method:
A common method for preparing 1,2-bis (di-tert-butylphosphine) ethane is to react sodium tert-butyrate with p-dibromoethane, and then react with tri-tert-butylphosphine to obtain the product.
Safety Information:
1,2-bis (di-tert-butylphosphino) ethane has low toxicity, but it still needs proper safety operation. When using, avoid contact with skin and eyes, and operate in a well-ventilated place. It should be kept away from ignition and heat sources and stored in an airtight container.
Nature:
1,2-Bis (di-tert-butylphosphino) ethane is a colorless liquid with a special smell. It is soluble in common organic solvents such as ethanol, acetone and dimethylformamide. It is a non-volatile liquid at room temperature and has a relatively low vapor pressure.
Use:
1,2-Bis (di-tert-butylphosphine) ethane is an important organic synthesis reagent, which is widely used in organic synthesis reactions. It can be used as a ligand in the catalyst system for hydrogenation, rearrangement, selective reduction and other reactions in organic synthesis. It can also be used in the synthesis of phosphinone compounds and transition metal phosphine complexes.
Preparation Method:
A common method for preparing 1,2-bis (di-tert-butylphosphine) ethane is to react sodium tert-butyrate with p-dibromoethane, and then react with tri-tert-butylphosphine to obtain the product.
Safety Information:
1,2-bis (di-tert-butylphosphino) ethane has low toxicity, but it still needs proper safety operation. When using, avoid contact with skin and eyes, and operate in a well-ventilated place. It should be kept away from ignition and heat sources and stored in an airtight container.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 1,2-Bis(di-tert-butylphosphino)ethane Request for quotation
CAS: 107783-62-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 107783-62-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 1,2-Bis(di-tert-butylphosphino)ethane Visit Supplier Webpage Request for quotation
CAS: 107783-62-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 107783-62-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 1,2-Bis(di-tert-butylphosphino)ethane Request for quotation
CAS: 107783-62-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 107783-62-0
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 1,2-Bis(di-tert-butylphosphino)ethane Visit Supplier Webpage Request for quotation
CAS: 107783-62-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 107783-62-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

