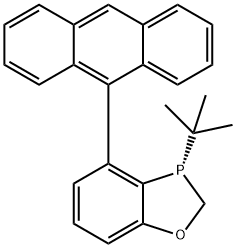
(S)-4-(9-Anthracenyl)-3-(tert-butyl)-2,3-dihydro-1,3-benzoxaphosphole
(S)-4-(anthracen-9-yl)-3-(te rt-butyl)-2,3-dihydrobenzo [d][1,3]oxaphosphole
CAS: 1807740-34-6
Molecular Formula: C25H23OP
(S)-4-(9-Anthracenyl)-3-(tert-butyl)-2,3-dihydro-1,3-benzoxaphosphole - Names and Identifiers
(S)-4-(9-Anthracenyl)-3-(tert-butyl)-2,3-dihydro-1,3-benzoxaphosphole - Physico-chemical Properties
| Molecular Formula | C25H23OP |
| Molar Mass | 370.42 |
| Boling Point | 525.7±50.0 °C(Predicted) |
| Appearance | crystal |
| Color | light yellow |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Sensitive | air sensitive, store cold |
(S)-4-(9-Anthracenyl)-3-(tert-butyl)-2,3-dihydro-1,3-benzoxaphosphole - Introduction
(S)-4-(anthracen-9-yl)-3-(te rt-butyl)-2,3-dihydrobenzo [d][1,3]oxaphosphole is an organic compound with the following properties:
1. Appearance: Usually a white crystalline solid.
2. Melting Point: about 120-125 ° C.
3. Molecular formula: C26H23OP.
4. Molecular weight: about 380g/mol.
5. Solubility: Soluble in most organic solvents, such as alcohols, ethers, etc.
The main uses of this compound include the following:
1. Catalyst: Phosphine-pentane can be used as chiral phosphine ligand to participate in the Catalyst System in organic synthesis reaction, such as asymmetric hydrogenation reaction, asymmetric nucleophilic substitution reaction.
2. Coordination chemistry: Phosphine-pentane yoke can form complexes with transition metals, which are widely used in organic metal chemical reactions, such as metal-catalyzed coupling reactions, Free Radical Polymerization reactions, etc.
3. Drug research: Phosphopentose and its derivatives are used in some drug research fields, such as the evaluation and analysis of the biological activity of anti-cancer drugs.
The preparation method of the phosphine-pentyl yoke is generally carried out by an organic synthetic chemical reaction. The specific method may include the reaction of benzophosphine oxide and Tri-tert-butyl phosphite to generate (S)-4-(anthracen-9-yl)-3-(te rt-butyl)-2,3-dihydrobenzo [d][1,3]oxaphosphole.
Regarding safety information, the phosphine yoke is a chemical, and the following safety precautions should be paid attention to when using it:
1. Avoid direct contact with skin and eyes, use to wear appropriate protective gloves and glasses.
2. During the operation, maintain good ventilation conditions to avoid inhaling the dust or vapor of the compound.
3. Storage, should be placed in a dry, cool place, away from the fire and oxidant.
4. In the treatment and disposal, follow the relevant local regulations and guidelines, pay attention to prevent its pollution to the environment.
1. Appearance: Usually a white crystalline solid.
2. Melting Point: about 120-125 ° C.
3. Molecular formula: C26H23OP.
4. Molecular weight: about 380g/mol.
5. Solubility: Soluble in most organic solvents, such as alcohols, ethers, etc.
The main uses of this compound include the following:
1. Catalyst: Phosphine-pentane can be used as chiral phosphine ligand to participate in the Catalyst System in organic synthesis reaction, such as asymmetric hydrogenation reaction, asymmetric nucleophilic substitution reaction.
2. Coordination chemistry: Phosphine-pentane yoke can form complexes with transition metals, which are widely used in organic metal chemical reactions, such as metal-catalyzed coupling reactions, Free Radical Polymerization reactions, etc.
3. Drug research: Phosphopentose and its derivatives are used in some drug research fields, such as the evaluation and analysis of the biological activity of anti-cancer drugs.
The preparation method of the phosphine-pentyl yoke is generally carried out by an organic synthetic chemical reaction. The specific method may include the reaction of benzophosphine oxide and Tri-tert-butyl phosphite to generate (S)-4-(anthracen-9-yl)-3-(te rt-butyl)-2,3-dihydrobenzo [d][1,3]oxaphosphole.
Regarding safety information, the phosphine yoke is a chemical, and the following safety precautions should be paid attention to when using it:
1. Avoid direct contact with skin and eyes, use to wear appropriate protective gloves and glasses.
2. During the operation, maintain good ventilation conditions to avoid inhaling the dust or vapor of the compound.
3. Storage, should be placed in a dry, cool place, away from the fire and oxidant.
4. In the treatment and disposal, follow the relevant local regulations and guidelines, pay attention to prevent its pollution to the environment.
Last Update:2024-04-10 22:41:03
Supplier List
Spot supply
Product Name: (S)-4-(anthracen-9-yl)-3-(tert-butyl)-2,3-dihydrobenzo[d][1,3]oxaphosphole Visit Supplier Webpage Request for quotationCAS: 1807740-34-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-4-(anthracen-9-yl)-3-(tert-butyl)-2,3-dihydrobenzo[d][1,3]oxaphosphole Request for quotation
CAS: 1807740-34-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 1807740-34-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: (S)-AntPhos Visit Supplier Webpage Request for quotation
CAS: 1807740-34-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1807740-34-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-4-(anthracen-9-yl)-3-(tert-butyl)-2,3-dihydrobenzo[d][1,3]oxaphosphole Visit Supplier Webpage Request for quotationCAS: 1807740-34-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-4-(anthracen-9-yl)-3-(tert-butyl)-2,3-dihydrobenzo[d][1,3]oxaphosphole Request for quotation
CAS: 1807740-34-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 1807740-34-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: (S)-AntPhos Visit Supplier Webpage Request for quotation
CAS: 1807740-34-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1807740-34-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


