tedizolid
tedizolid
CAS: 856866-72-3
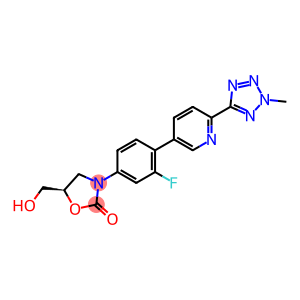
Molecular Formula: C17H15FN6O3
tedizolid - Names and Identifiers
tedizolid - Physico-chemical Properties
| Molecular Formula | C17H15FN6O3 |
| Molar Mass | 370.34 |
| Density | 1.57 |
| Melting Point | 201 °C |
| Boling Point | 614.5±65.0 °C(Predicted) |
| Solubility | DMSO (Slightly, Heated), Water (Slightly, Heated) |
| Appearance | Solid |
| Color | White to Off-White |
| pKa | 14.05±0.10(Predicted) |
| Storage Condition | Refrigerator |
| Use | Torezolid (TR-701; tedizolid) is a reversible inhibitor of human MAO-A and MAO-B with activity against gram-positive bacteria. |
tedizolid - Risk and Safety
| HS Code | 29339900 |
tedizolid - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.7 ml | 13.501 ml | 27.002 ml |
| 5 mM | 0.54 ml | 2.7 ml | 5.4 ml |
| 10 mM | 0.27 ml | 1.35 ml | 2.7 ml |
| 5 mM | 0.054 ml | 0.27 ml | 0.54 ml |
Last Update:2024-01-02 23:10:35
tedizolid - Reference Information
| Background | Tedizolid phosphate is a second-generation oxazolidinone antibiotic developed by Pharmaceutical Dong-A, cubist Pharmaceuticals and Bayer are authorized for commercial development. Sivextro is intended for the treatment of serious or life-threatening infections and its marketing application has been granted the qualification of a Qualified Infectious Disease Product (QIDP) and received accelerated review by the FDA. Sivextro's QIDP qualification gives the drug an additional 5 years of market exclusivity in addition to the market exclusivity conferred by the Food, Drug, and Cosmetic Act. The safety and efficacy of Sivextro were evaluated in two clinical trials involving 1315 adult patients with ABSSSI. Subjects were randomized to Sivextro or another approved antibacterial drug, linezolid, for the treatment of acute bacterial skin and skin structure infections (ABSSSI). The results show that the clinical effect of linezolid is equivalent to that of linezolid. The adverse reactions in gastrointestinal tract and thrombocytopenia are less than that of linezolid, and the incidence of drug resistance is lower. Some trials have shown that the resistance of the drug is also better than that of vancomycin. The most common side effects confirmed in clinical trials were Nausea, Head Pain, Diarrhea, Vomit and dizziness. The safety and efficacy of Sivextro has not been evaluated in patients with reduced white blood cell levels (neutropenia), so alternative therapies should be considered. Sivextro is marketed by Cubist Pharmaceuticals, located in Rosenberg, Massachusetts. Intellectual property status: patent ZL200480037612.2 for compound, which expires on 2024.12.17. This information was edited by chemical book Xiaonan (2015-08-14). |
| Introduction | terdizolamide is a second generation oxazolidinone antibiotic, which is an analog of linezolid, is a protein synthesis inhibitor that acts on the bacterial ribosomal 50S subunit to inhibit bacterial protein synthesis. Compared with linezolid, the efficacy of the two is basically the same, but linezolid needs to be taken twice a day for 10 consecutive days, while terezolid only needs to be taken once a day, the drug was taken for 6 consecutive days. On June 20, 2014, the U. S. FDA approved a new antibacterial agent, terdizolamide (trade name: Sivextro), for the treatment of skin infections in adult patients. Sivextro is approved for the treatment of certain sensitive bacteria such as Staphylococcus aureus (including methicillin-resistant strains and methicillin-sensitive strains), acute bacterial skin and skin tissue infections caused by various streptococci and Enterococcus faecalis (ABSSSI). Sivextro was administered intravenously and orally. |
| indication | Tedizolid Phosphate (tedizolamide) is an oxazolidinone compound, for acute bacterial skin and skin structure infections caused by sensitive strains of the following Gram-positive bacteria: Staphylococcus aureus (including methicillin-resistant and methicillin-sensitive strains), Streptococcus pyogenes, * Streptococcus lactis, angina streptococcus group (including angina Streptococcus, Streptococcus intermediate, streptococcus group), Enterococcus faecalis. |
| Intellectual Property Status | (I) administrative protection, new drug protection, new drug monitoring period , bayer Medical has submitted a new drug application to the CFDA. (B) domestic patents 1. Compound patent East Asia Pharmaceuticals has applied for a patent for the compound in China, with the application number of ZL200480037612.2, with the application date of 17 December 2004, and the protection period expires on 2024.12.17. Three additional divisional applications were subsequently submitted, with application numbers of 201010508824.1 (formulation),201110304983.4, and 201210155386.4, respectively. on the 9th of, a patent application for the preparation method was made by the company of treus therapeutics, with the application number of 200980140144.4. 2. Patent of crystal form on the 3rd of, the Telus therapeutics company applied for a patent of crystal form of free acid with the application number of 201080014363.0. The crystalline form I of the free acid is disclosed to have the advantage of being more stable than the disodium salt and having no hygroscopicity. 3. Formulation composition patent The above-mentioned patent 200480037612.2 and its divided application patent disclose tedizolamide powder, tablet, capsule and injection formulation. The above-mentioned patent no. 201080014363.0 discloses the formulation of tedizolamide tablets and lyophilized formulations. |
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Torezolid Request for quotation
CAS: 856866-72-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 856866-72-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Torezolid Request for quotation
CAS: 856866-72-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 856866-72-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Tedizolid Visit Supplier Webpage Request for quotationCAS: 856866-72-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




