methane-D3
methane-D3
CAS: 676-80-2
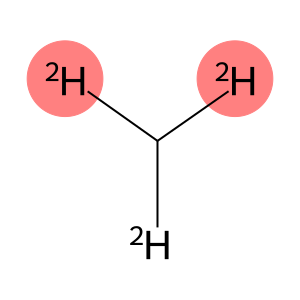
Molecular Formula: CHD3
methane-D3 - Names and Identifiers
| Name | methane-D3 |
| Synonyms | methane-D3 METHANE-D3 Deuterium methyl Methane-D3 (gas) trideuteriomethane |
| CAS | 676-80-2 |
| InChI | InChI=1/CH4/h1H4/i1D3 |
methane-D3 - Physico-chemical Properties
| Molecular Formula | CHD3 |
| Molar Mass | 19.06 |
| Melting Point | −183°C(lit.) |
| Boling Point | −161°C(lit.) |
| Flash Point | -188℃ |
| Vapor Presure | 205285.6mmHg at 25°C |
| Vapor Density | 0.55 (vs air) |
methane-D3 - Risk and Safety
| Hazard Symbols | F+ - Highly flammable |
| Risk Codes | 12 - Extremely Flammable |
| Safety Description | S16 - Keep away from sources of ignition. S38 - In case of insufficient ventilation, wear suitable respiratory equipment. |
| UN IDs | UN 1971 2.1 |
| WGK Germany | 3 |
methane-D3 - Introduction
methane-D3(methane-D3) refers to an isotopically labeled methane molecule in which three protons in a hydrogen atom are replaced by deuterium (D). Its chemical formula is CD3H.
Nature:
The physical properties of methane-D3 are similar to ordinary methane, which is a colorless, tasteless and odorless gas. Its density is smaller than the air, insoluble in water, soluble in organic solvents, flammable.
Use:
methane-D3 are commonly used in nuclear magnetic resonance (NMR) experiments as solvents and reference materials. NMR is a commonly used analytical technique that can solve the structure, composition, and dynamics of compounds by observing the behavior of molecules in a magnetic field. methane-D3 as an NMR solvent can provide a solvent environment of high purity and high stability.
Preparation Method:
methane-D3 can be prepared by reacting deuterium gas with chlorinated methane. One common method is to react chlorinated methane with deuterium gas in the gas phase in the presence of a catalyst. This process allows some or all of the hydrogen atoms in the methane molecule to be replaced by deuterium, resulting in methane-D3.
Safety Information:
methane-D3 is unstable under general temperature and pressure and easy to burn. Keep away from fire and high temperature environment when used. At the same time, methane-D3 may have a suffocating effect, and attention should be paid to ventilation conditions to avoid prolonged exposure to high concentrations. In addition, for chemical laboratories, operators should wear appropriate protective equipment such as gloves, safety glasses and laboratory goggles to ensure safety. In the use and storage process, but also properly manage the use and handling of related substances, follow the relevant safety procedures.
Nature:
The physical properties of methane-D3 are similar to ordinary methane, which is a colorless, tasteless and odorless gas. Its density is smaller than the air, insoluble in water, soluble in organic solvents, flammable.
Use:
methane-D3 are commonly used in nuclear magnetic resonance (NMR) experiments as solvents and reference materials. NMR is a commonly used analytical technique that can solve the structure, composition, and dynamics of compounds by observing the behavior of molecules in a magnetic field. methane-D3 as an NMR solvent can provide a solvent environment of high purity and high stability.
Preparation Method:
methane-D3 can be prepared by reacting deuterium gas with chlorinated methane. One common method is to react chlorinated methane with deuterium gas in the gas phase in the presence of a catalyst. This process allows some or all of the hydrogen atoms in the methane molecule to be replaced by deuterium, resulting in methane-D3.
Safety Information:
methane-D3 is unstable under general temperature and pressure and easy to burn. Keep away from fire and high temperature environment when used. At the same time, methane-D3 may have a suffocating effect, and attention should be paid to ventilation conditions to avoid prolonged exposure to high concentrations. In addition, for chemical laboratories, operators should wear appropriate protective equipment such as gloves, safety glasses and laboratory goggles to ensure safety. In the use and storage process, but also properly manage the use and handling of related substances, follow the relevant safety procedures.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: METHANE-D3 Request for quotation
CAS: 676-80-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 676-80-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: METHANE-D3 Request for quotation
CAS: 676-80-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 676-80-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History
