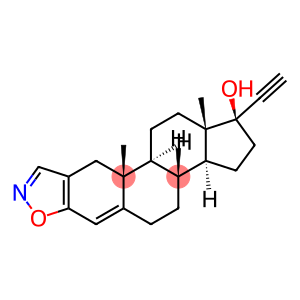
danazol
danazol
CAS: 17230-88-5
Molecular Formula: C22H27NO2
danazol - Names and Identifiers
danazol - Physico-chemical Properties
| Molecular Formula | C22H27NO2 |
| Molar Mass | 337.46 |
| Density | 1.0909 (rough estimate) |
| Melting Point | 224.4-226.80C |
| Boling Point | 473.76°C (rough estimate) |
| Specific Rotation(α) | D25 +7.5° (ethanol); D25 +21.9° (chloroform) |
| Flash Point | 243.003°C |
| Water Solubility | Partly soluble in water. Soluble in chloroform (25 mg/ml), acetone, acetonitrile, and ethanol. 17beta-Hydroxy-2,4,17a-pregnadien-20-yno[2,3-d]isoxazole and 2,4,17a-Pregnadien-20-yno[2,3-d]isoxazol-17- |
| Solubility | Acetonitrile (Slightly), Chloroform (Slightly), Ethanol (Slightly), Methanol (Sl |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Solid |
| Color | White to Pale Yellow |
| pKa | 13.10±0.60(Predicted) |
| Storage Condition | -20°C Freezer |
| Refractive Index | 1.5614 (estimate) |
| Use | For the treatment of endometriosis and idiopathic thrombocytopenia |
danazol - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R63 - Possible risk of harm to the unborn child R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S22 - Do not breathe dust. S36 - Wear suitable protective clothing. S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | TU4157070 |
| HS Code | 29372900 |
danazol - Standard
Authoritative Data Verified Data
This product is 17a-pregnane-2, 4-dien-20-acetylino [2-3-d] isoxazol-17b-ol. The content of C22H27N02 shall be 97.0% ~ 103.0% calculated as dry product.
danazol - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline or crystalline powder.
- This product is soluble in chloroform, soluble in acetone, slightly soluble in ethanol, insoluble in water.
specific rotation
take this product, precision weighing, plus three gas methane dissolution and quantitative dilution of about 10 mg per 1 ml of the solution, according to the law (General 0621), the specific rotation was 21 ° to 27 °.
danazol - Differential diagnosis
Authoritative Data Verified Data
- take about 2mg of this product, add 5ml of ethanol to dissolve, add 2 drops of silver nitrate test solution to generate white precipitate.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 147).
danazol - Exam
Authoritative Data Verified Data
chloride
take this product l. Add 30ml of water, shake to disperse evenly, filter, take 15ml of filtrate, check according to law (General rule 0801), and standard sodium chloride solution 4.0ml of the control solution should not be more concentrated (0.008%).
sulfate
take this product l.Og, add water 30ml, shake to disperse evenly, filter, take filtrate 15ml, check according to law (General rule 0802), compared with the control solution made of standard potassium sulfate solution 4.0ml, not more concentrated (0.08%).
Related substances
take this product, add mobile phase to dissolve and dilute to make a solution containing about 2mg per 1ml as a test solution; Take 1ml for precision measurement and put it in a 100ml measuring flask, dilute to the scale with the mobile phase, shake, and serve as a control solution. According to the chromatographic conditions under the content determination item, 10 u1 of the test solution and 10 u1 of the control solution are respectively injected into the liquid chromatograph, and the chromatogram is recorded to 2 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.5 times (0.5%) of the area of the main peak of the control solution; the sum of each impurity peak area shall not be greater than the main peak area of the control solution (1.0%).
loss on drying
take this product, at 60°C under reduced pressure drying to constant weight, weight loss should not exceed 1.0% (General rule 0831).
danazol - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with eighteen alkyl silane was used as a filler; Acetonitrile-methanol-water (4:4:3) was used as a mobile phase; The detection wavelength was 270mn. The number of theoretical plates is not less than 2500 based on the danazol peak.
assay
take this product, precision weighing, adding mobile phase dissolution and quantitative dilution to make a solution containing about 0.2mg per 1 ml, as a test solution, take the LOL injection liquid chromatograph, the chromatogram was recorded; Another reference substance of Danazol was taken for determination by the same method. According to the external standard method to calculate the peak area, that is.
danazol - Category
Authoritative Data Verified Data
gonadotropin inhibitory drugs.
danazol - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
danazol - Danazol Capsules
Authoritative Data Verified Data
This product contains Danazol (C22H27N02) should be labeled the amount of 90.0% ~ 110.0%.
identification
- take the content of this product, add the appropriate amount of three gas methane extraction, filtration, filtrate evaporation, extract according to danazol identification (1) test, showed the same reaction.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take an appropriate amount of the contents of this product (about 0402 mg equivalent to danazol), add of chloroform, grind and dissolve, filter, evaporate the filtrate in water bath, dry the residue under reduced pressure, and determine it according to law (general rule). The infrared absorption spectrum of this product should be consistent with the spectrum of the control (Spectrum set 147 Figure).
examination
- the contents under the item of difference in loading amount of related substances should be mixed evenly, accurately weighed to take appropriate amount, dissolved with mobile phase and diluted to prepare a solution containing about 2mg of danazol per 1 ml, and filtered, the filtrate was taken as a test solution and tested according to the method under danazol related substances. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.75 times (0.75%) of the area of the main peak of the control solution, the sum of each impurity peak area shall not be greater than 1.5 times (1.5%) of the main peak area of the control solution.
- dissolution of this product, according to the dissolution and release determination method (General 0931 second method), with 0.lmol/L hydrochloric acid solution-isopropyl alcohol (3:2)l000ml as the dissolution medium, the rotation speed is 80 rpm, according to the law, after 30 minutes, take 25ml solution filtration, the appropriate amount of continued filtrate was accurately taken and diluted quantitatively with dissolution medium to make a solution containing about 20 waves of danazol per 1 ml, the dissolution medium was added to dissolve and quantitatively dilute to prepare a solution containing about 20 WW per 1 ml. Take the above two kinds of solution, according to UV-visible spectrophotometry (General rule 0401), at the wavelength of 286nm absorbance were measured, calculate the amount of each dissolution, limit is 70% of the label amount, shall comply with regulations,
- others should comply with the relevant provisions under the capsule (General 0103).
Content determination
take the contents under the difference of loading amount, mix evenly, weigh an appropriate amount (about 20mg equivalent to danazol) accurately, put it in a 100ml measuring flask, and add an appropriate amount of mobile phase, sonicate danazol, cool, dilute to the scale with mobile phase, shake, filter, take the filtrate as the test solution, and determine it according to the method under the item of danazol content determination, that's right.
category
syndanazole.
specification
(1)0.lg (2)0.2g
storage
light shielding, sealed storage.
Supplier List
CAS: 17230-88-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 17230-88-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 17230-88-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 17230-88-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501

