carprofen
carprofen
CAS: 53716-49-7
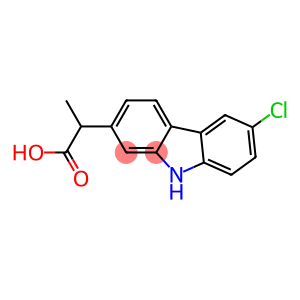
Molecular Formula: C15H12ClNO2
carprofen - Names and Identifiers
| Name | carprofen |
| Synonyms | carprofen CARPROFEN Carprofen (200 mg) Carprofen (200 mg) (AS) methylcarbazole-2-acetic acid 2-(6-chloro-9H-carbazol-2-yl)propanoic acid 6-CHLORO-ALPHA-METHYL-9H-CARBAZOLE-2-ACETIC ACID Carprofen,6-Chloro-α-methyl-9H-carbazole-2-acetic acid |
| CAS | 53716-49-7 |
| EINECS | 258-712-4 |
| InChI | InChI=1/C15H12ClNO2/c1-8(15(18)19)9-2-4-11-12-7-10(16)3-5-13(12)17-14(11)6-9/h2-8,17H,1H3,(H,18,19) |
| InChIKey | PUXBGTOOZJQSKH-UHFFFAOYSA-N |
carprofen - Physico-chemical Properties
| Molecular Formula | C15H12ClNO2 |
| Molar Mass | 273.71 |
| Density | 1.2011 (rough estimate) |
| Melting Point | 186-1880C |
| Boling Point | 509.1±35.0 °C(Predicted) |
| Flash Point | 261.7°C |
| Solubility | DMSO: soluble20mg/mL, clear |
| Vapor Presure | 3.45E-11mmHg at 25°C |
| Appearance | powder |
| Color | white to beige |
| Merck | 14,1862 |
| pKa | 4.84±0.30(Predicted) |
| Storage Condition | Sealed in dry,2-8°C |
| Refractive Index | 1.5200 (estimate) |
| Physical and Chemical Properties | White or off-white crystalline powder. Melting point 197-198 °c. |
| Use | Used for pain relief of rheumatoid arthritis, osteoarthritis, acute gout, extra-articular rheumatism, etc |
| In vitro study | Carprofen(S and R stereoisomers) inhibits canine COX2 with an IC50 of 0.102 microM. The main effect is to inhibit the S enantiomer (IC50 is 0.0371 microM), which is more than 200 times higher than that of the R enantiomer (IC50 is 5.97 microM). Carprofen binds to human serum albumin (HSA) by fluorescence and equilibrium dialysis (ED) methods with two binding constants [K1=5.1 μm (fluorescence) and 3.7 μm (ED), K2 = 37 μm (fluorescence) and 13 μm (ED)]. Carprofen mainly binds to site II,Benzodiazepine site, the low affinity site of Carprofen is site I,Warfarin site, the carboxyl group of Carprofen plays an important role in the high affinity binding of Carprofen and HSA. |
| In vivo study | Before surgery, Carprofen had lower pain scores than other groups, and was significantly effective in canines treated at 2 hours after extubation. Treatment of dogs with Carprofen (4 mg/kg) resulted in significantly higher maximum plasma concentrations, area under the curve to the last data point, and the area under the curve at the first moment is also up to the last data point. Canines treated with Carprofen (4 mg/kg) had a slightly better analgesic effect than Pethidine and produced less sedation. Carprofen treatment of dogs for 18 hours provided good analgesia without adverse side effects. Carprofen greatly increases the speed of the lame bird. |
carprofen - Risk and Safety
| Risk Codes | R25 - Toxic if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| UN IDs | UN 2811 |
| WGK Germany | 3 |
| RTECS | FE3180000 |
| HS Code | 29339900 |
| Hazard Class | 6.1 |
| Packing Group | III |
| Toxicity | LD50 orally in mice: 400 mg/kg (Berger, Corraz) |
carprofen - Reference
| Reference Show more | 1. Song Yanbo, Zhao Guishan, Ru Xiaofei, Hou Xiaolin, Mengzhou, Li Xinjie, Meng Qingrong, Wang Xiaodong. Preparation of artificial antigen and polyclonal antibody of indomethacin [J]. Animal husbandry and veterinary medicine 2019 51(10):44-48. |
Supplier List
Spot supply
Product Name: Carprofen Visit Supplier Webpage Request for quotationCAS: 53716-49-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Carprofen Request for quotation
CAS: 53716-49-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 53716-49-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: carprofen Request for quotation
CAS: 53716-49-7
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 53716-49-7
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Multiple SpecificationsSpot supply
Product Name: 6-Chloro-a-Methyl-9H-Carbazole-2-Acetic Acid Visit Supplier Webpage Request for quotationCAS: 53716-49-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Carprofen Visit Supplier Webpage Request for quotationCAS: 53716-49-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Carprofen Request for quotation
CAS: 53716-49-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 53716-49-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: carprofen Request for quotation
CAS: 53716-49-7
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 53716-49-7
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Multiple SpecificationsSpot supply
Product Name: 6-Chloro-a-Methyl-9H-Carbazole-2-Acetic Acid Visit Supplier Webpage Request for quotationCAS: 53716-49-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



